Advanced pharmaceutical bulletin. 14(4):819-835.
doi: 10.34172/apb.39216
Mini Review
The Role of Estrogen in Brain MicroRNAs Regulation
Peyvand Bahramiazar Conceptualization, Investigation, Resources, Visualization, Writing – original draft, 1 
Naseh Abdollahzade Investigation, Resources, Software, Visualization, Writing – review & editing, 2, * 
Bakhtyar Tartibian Project administration, Supervision, 3
Naser Ahmadiasl Conceptualization, Project administration, Supervision, 1
Fakhreddin Yaghoob Nezhad Investigation, Project administration, Resources, Validation, Visualization, Writing – original draft, Writing – review & editing, 4, * 
Author information:
1Department of Physiology, Tabriz University of Medical Sciences, Tabriz, Iran.
2Neurophysiology Research Center, Cellular and Molecular Medicine Institute, Urmia University of Medical Sciences, Urmia, Iran.
3Department of Exercise Physiology, Faculty of Physical Education and Sport Sciences, Allameh Tabataba’i University, Tehran, Iran.
4TUM School of Medicine and Health, Technical University of Munich, Germany.
Abstract
Purpose:
This review aims to elucidate the role of estrogen-sensitive microRNAs (miRNAs) in modulating brain functions and disorders, highlighting the protective effects of estrogen on the central nervous system.
Methods:
A comprehensive literature review was conducted, examining the relationship between estrogen, miRNAs, and cognitive health. The study focused on experimental data comparing cognitive impairments between genders and the mechanisms of estrogen’s effects on brain function.
Results:
Cognitive impairments are less prevalent in women of reproductive age compared to men, indicating estrogen’s neuroprotective role. Estrogen modulates gene expression through specific receptors, while miRNAs regulate approximately 30% of protein-coding genes in mammals. These miRNAs play critical roles in synaptic plasticity and neuronal survival. The review identifies several estrogen-sensitive miRNAs and their potential involvement in brain disorders.
Conclusion:
The interplay between estrogen and miRNAs offers valuable insights into the molecular mechanisms underlying cognitive health and disease. Understanding these relationships may lead to novel therapeutic strategies for addressing various brain disorders, particularly those associated with hormonal changes and aging.
Keywords: miRNA, Estrogen, Brain, Aging, Ovariectomy
Copyright and License Information
©2024 The Author (s).
This is an Open Access article distributed under the terms of the Creative Commons Attribution (CC BY), which permits unrestricted use, distribution, and reproduction in any medium, as long as the original authors and source are cited. No permission is required from the authors or the publishers.
Funding Statement
None.
Introduction
Brain and neurological disorders are among the major causes of mortality and disability throughout the world. According to recent investigations, the incidence of these diseases will increase in the next decades.1 The incidence of diabetes, psychiatric problems, hypertension, and degenerative brain diseases, such as dementia and stroke, is more frequently seen in elderly people. Lack of effective treatment leads to turning these diseases into a major problem.2 Although it is not clear how age contributes to these diseases, studies have shown cognitive complaints are common in women, who are transitioning through menopause.3 It is important, therefore, to understand the effects of normal ovarian aging and menopause on the CNS. Both experimental and clinical data have revealed the benefits of estrogen in the brain.4 Estrogen has neurotrophic and neuroprotective properties and is necessary for conserving learning and memory.5 It has also been shown estradiol attenuates reactive oxygen species release,6 inhibits the production of macrophage cytokines, decreases expression of the inducible isoform of nitric oxide synthases, and increases anti-inflammatory signaling pathways.7 Various studies have indicated that ovariectomy (OVX) increases brain damage and neurodegenerative processes as a result of decreasing estrogen levels.8 Furthermore, hormonal therapy restores the estradiol levels and recovers brain damage in ovariectomized animals.9 It is proven that estrogen controls various signaling pathways through its receptors,10 and subsequently, regulates the expression of microRNAs (miRNAs) in most tissues such as the brain.11 miRNAs regulate protein translation by binding their target messenger RNAs (mRNAs) and inhibiting the translating or degrading mRNA molecules.12 Emerging evidence shows that miRNAs have nuclear functions at the transcriptional level in terms of regulating gene expression.13 Thus, miRNA-mediated regulation is now considered one of the most important post-transcriptional gene regulation mechanisms and regulates more than 30% of mammal genes, including important roles in human physiology, aging, and CNS disorders.14 Even though many studies have suggested miRNAs as crucial epigenetic regulators of brain function, their role in regulating estrogen in brain disorders is not fully understood. This review aims to identify current knowledge of the role of estrogens in brain function with a special focus on the miRNAs that are regulated by estrogens.
MiRNA functions and processing overview
MiRNAs are a class of small (~20-22 nucleotide) non-coding RNAs that appear to have been extensively researched from the time of their discovery by Ambros,15 and Ruvkun,16 in Caenorhabditis elegans and Baulcombe,17 in plants and their prominent role in cardiovascular disease, diabetes, cancers and aging disorders has been shown.
Recent evidence indicates that miRNAs play important roles in regulating various cellular processes including translation of select mRNAs, apoptosis, differentiation, and replication.18-20 The human genome contains around 2588 mature miRNAs.21 About 50% of miRNAs are intragenic and most of them are found within the 50 introns of host genes.22 In studies, it is estimated that 1-4% of the human genome is made up of miRNAs, and one miRNA can regulate about 200 mRNAs,23 and since they are involved in many human biological processes, their regulation may be disturbed. lead to widespread disorders in the body, so that about 70 miRNA-related diseases have been reported (http://cmbi.bjmu.edu.cn/hmdd). It is likely that the reports of these cases will increase over time. The miRBase reference repository currently contains information on 1917 human precursors and 2656 mature miRNAs (version 22).24 that this figure is increasing day by day, although a number of false positive cases have also been reported among them, which the reference databases try to minimize these cases, so that there is a significant reduction of miRNAs with high confidence in the new versions of miRBase Is. However, the quality of relevant miRNA databases ultimately always depends on the availability of highly reliable positive and negative training sets, i.e., miRNAs validated by appropriate experimental methods. and requires global definition of criteria.25,26 However, based on experiments listed in miRBase, there is evidence of miRNA expression by NB for only 3.6% of all human miRNAs listed in miRBase V22.27-29 And recently, the studies of Alles et al,30 who used an experimental method with high efficiency and reliability and in different tissues, data showed 2300 mature human miRNAs, 50% of them were also included in miRBase V22. Although this number cannot be final, based on the data presented, it can represent a high estimate of what we can expect as the final extent of the real human miRNome.
For the first time, miRNA was discovered in 1993 and seemed to be involved in C. elegans developmental timing (Elegans heterochronic gene lin-4, that encodes small RNAs to lin-14 with antisense complementarity). It has been understood that miRNAs are expressed in every eukaryotic organism and are involved in almost all cellular functions. miRNAs inhibit the translation of their targets or mediate mRNA decay.31 miRNA affects mRNAs and could eliminate the 3_ polyA tail as well as 5_cap, which in turn degrades and destabilizes this mRNA.32 When miRNAs hinder translation without mRNA decay, they often avoid the initiation stage of translation but can influence translation postinitiation.33
Approximately 60-100 nt pri-miRNAs are transcribed by RNA polymerase II.34 Drosha (an RNAse III endonuclease) processes Pri-miRNAs, which are recruited co-transcriptionally.35 The Drosha complex is linked to 20 polypeptides, known as the Drosha microprocessor complex.36 The Drosha cofactor DiGeorge critical region 8 (DGCR8) is a double-stranded RNA binding domain protein (dsRBD), which is necessary to cleave pri-miRNA into 60-70 nt imperfect hairpin precursor-miRNAs (pre-miRNAs). Drosha can be stabilized by DGCR8 and controls the levels of DGCR8 by cleaving DGCR8 mRNA.37
The DEAD-box RNA helicases p68 and p72 are the components of the Drosha complex that have roles in processing a subset of miRNAs.38 Exportin-5 and Ran-GTP export Pre-miRNAs to the cytoplasm. miRNAs are processed by the Dicer complex inside the cytoplasm. Dicer (a cytoplasmic RNAse III) removes the loop structure of pre-miRNA to create the mature ~22 nt miRNA duplex.39 Dicer transfers miRNA to Argonaute proteins (Ago1, Ago2, Ago3, and Ago4), TRBP, and PACT in the RNA-induced silencing complex (RISC).40 Ago proteins change the miRNA duplexes and form products like single-stranded miRNA-5p and miRNA-3p. Complementary binding of the mature miRNA to the 3-untranslated region of a target mRNA, and its association with the RISC, result in translational mRNA degradation or repression41 (Figure 1).
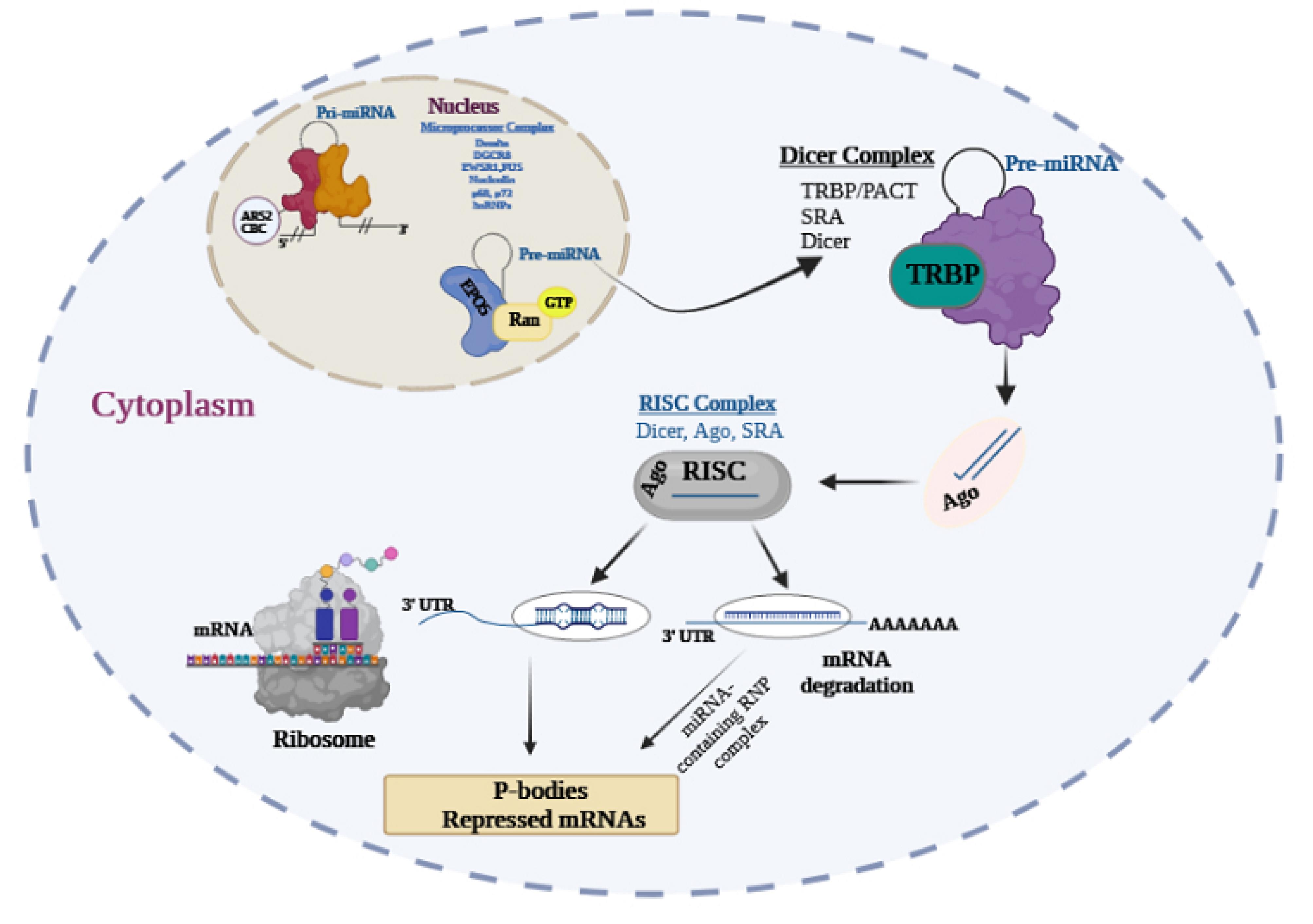
Figure 1.
Processing pathway of miRNA: The figure reveals the processing of miRNA. miRNAs are converted to pri-miRNAs by RNA polymerase II, which is processed into precursors – pre-miRNAs by the Drosha microprocessor complex, which contains DGCR8. The p72 and DEAD-box RNA helicases p68 belong to the Drosha complex. Exportin-5 recognizes Ran-GTP and Pre miRNAs with GTP hydrolysis sent to the cytoplasm. Pre-miRNA is processed to mature miRNA by the Dicer complex in the cytoplasm. The TRBP/PACT complex interconnects with Dicer and stabilizes it, then transfers miRNA to Ago proteins in the RISC. miRNA -by binding to the 30 untranslated regions - conductors the RISC complex to target mRNAs and lead to inhibition of the translation. The Silent mRNA degradation occurs in P-bodies
.
Processing pathway of miRNA: The figure reveals the processing of miRNA. miRNAs are converted to pri-miRNAs by RNA polymerase II, which is processed into precursors – pre-miRNAs by the Drosha microprocessor complex, which contains DGCR8. The p72 and DEAD-box RNA helicases p68 belong to the Drosha complex. Exportin-5 recognizes Ran-GTP and Pre miRNAs with GTP hydrolysis sent to the cytoplasm. Pre-miRNA is processed to mature miRNA by the Dicer complex in the cytoplasm. The TRBP/PACT complex interconnects with Dicer and stabilizes it, then transfers miRNA to Ago proteins in the RISC. miRNA -by binding to the 30 untranslated regions - conductors the RISC complex to target mRNAs and lead to inhibition of the translation. The Silent mRNA degradation occurs in P-bodies
It has been discovered that more than 2000 miRNAs exist in human cells, most of these miRNAs are specific in some cells, tissues, or organs.42 They control many biological procedures, like cell differentiation and proliferation, growth control, apoptosis, and development.42,43
Studies have shown that a wide range of human diseases are related to changes in the expression of miRNAs, and in many cases these disorders are considered as a cause in the progression of diseases. Therefore, extensive efforts have been made to develop miRNA therapy, depending on For the purpose of treatment, these studies have been conducted on inhibitors and enhancers of miRNAs, and the most promising drugs that have been introduced so far are drugs such as: Regulus RG-101, which is an inhibitor of miR-122 and it is used to treat HCV infection,44 or drugs such as let-7, which are used to treat cancer diseases, or the use of miR-34 in cancer and cardiovascular diseases, as well as the use of miR-29 for fibrosis.45,46
Mechanisms for these therapeutic goals have been introduced, which fall into two general categories: therapeutic strategies for miRNA-decreasing diseases and therapeutic strategies for miRNA-enhancing diseases, which in the first category substitute a miRNA mimetic for diseases in which miRNAs are decreased. It is found that this imitation is usually a dsRNA duplex that is loaded in RISC or a stem loop precursor is loaded in RISC, but in the second category where the increase of miRNAs occurs in diseases, oligonucleotide inhibitors can be used. which, as an inhibitor, prevents the binding of miRNA to the target miRNAs by entering the RISC complex.45,47
Studies show that miRNAs are involved in regulating brain development and disorder.48 However, the practical treatment based on the use of microRNAs for neurological diseases has not yet been introduced, and most of the studies conducted have been aimed at proposing strategies and treatment solutions needed for this issue, so we also tried in this review. By examining the various therapeutic aspects of microRNAs and the role and beneficial effects of estrogen use, let’s give the researchers in this field a good view in choosing a more effective and efficient treatment. (Table 1)
Table 1.
The most abundant miRNAs in the brain
|
miRNA
|
Main findings
|
Cell lines
|
| miRNA-9 |
|
-
P19 Mouse cortical cells
-
SK-N-BE cells
|
| miRNA-124 |
-
A role for miR-124 in regulating the regeneration of a functional brain and visual system.51
-
Overexpression of miR-124 led to an enhancement of neuronal incorporation and differentiation of neural-specific exons in some genes.52
-
Increased levels of miR-124 in neuronal differentiation induced by retinoic acid.53
|
-
Planarian Schmidtea mediterranea
-
mouse neuroblastoma cell lines
-
human NTera2D1 and rodent P19D and Neuro2a (N2a) cells
|
| miRNA-128 |
-
mir-128 can inhibit the proliferation of glioma cells by negatively regulating one of its targets, E2F3a, which is highly expressed in glioma and important for cell cycle progression.54
-
miR-128 up-regulation inhibits DCX and Reelin expression and reduces neuroblastoma cell invasiveness and cell motility in cell lines.55
|
|
| miRNA-10 |
|
-
Human glioma A172, U87, LN229,U251, and neuroblastoma SHSY5Y
|
| miRNA- 29 |
|
|
| let-7b, let-7d |
|
-
The hypothalamus, hippocampus, occipital cortex, prefrontal cortex, and somatosensory cortex of male rats
|
| miRNA-29 |
|
|
| miRNA-132 |
-
The overexpression of miR-132 increased synaptic MMP-9 level provokes enlargement of the dendritic spine heads.59
-
miR-132 expression influences neuronal maturation via its effects on dendritic and spinogenesis arborization.60
|
-
Cultured hippocampal neurons from Fmr1 KO mice
-
CA3, CA1and GCL excitatory cell layers of the C57/Bl6 mice hippocampus
|
| miRNA-134 |
-
miR-134 Regulates Ischemia/Reperfusion Injury-Induced Neuronal Cell Death by Regulating CREB Signaling.61
-
miR-134 is a negative posttranscriptional regulator of GluA2 expression in Hippocampal Neurons.62
|
|
| miRNA-219 |
|
|
| miRNA-425 |
|
|
| miRNA-181 |
|
-
Human glioma cell lines, U251, U87 and TJ905
-
HEK-293 cells or Male Wistar rat brain parts
|
| miRNA-15 |
-
miR-15a is a target of PPAR- transrepression directly regulates BCL-2 and contributes to PPAR-mediated vascular protection against ischemia-like insults.67
-
miR‐15a regulates oxygen-glucose deprivation/reperfusion (OGD/R)‐induced neuronal injury by targeting BDNF.68
|
-
Cerebral cortex from adult male C57BL/6J mice
-
Primary cortical neurons of Sprague‐Dawley rats, 16‐18 days of age
|
| miRNA-200 |
|
|
| miRNA-125 |
-
miR-125b Overexpression led to an enhancement of neuronal proliferation and differentiation in neural stem/progenitor cells.58
-
miR-125a Increased neuronal differentiation levels induced by retinoic acid.57
|
|
| miRNA-106 |
|
|
| miRNA-210 |
|
|
| miRNA-324 |
|
|
| miRNA-138 |
|
|
| miRNA-338 |
|
|
| miRNA-339 |
|
|
Initial studies show that miRNAs are expressed and enriched in region-specific areas of the brain.76 miRNAs have important roles in different neurologic processes including neuronal development (miRNA-10, miRNA-9, miRNA-430),77 and neuronal cell maintenance (miRNA-29, miRNA-134).56,78 The most abundant miRNAs related to neurons include miRNA-124, miRNA-9, miRNA-128, miRNA-29, let-7, and miRNA-26.79,80,81
A miRNA that is especially expressed in the central nervous system is miRNA-124,82 and is predominantly expressed in post-mitotic neurons.83 During brain development, the expression of miR-124 increases.84 miRNA-124 regulates neuronal differentiation and maturation by the expression of small GTPase Ras homolog growth-related (RhoG),85 LIM/homeobox protein 2 (Lhx2),86 Rho-associated coiled-coil-containing protein kinase 1 (ROCK1),87 and cAMP response element-binding (CREB) protein.88 It has been shown recently that miR-124 regulates cognition and synaptic transmission by restricting the expression of early growth response gene 1 (Egr1).88
Another miRNA during brain development is miR-132, which regulates neuronal structure78 and can contribute to spine morphogenesis,89 arborization, and dendritic growth in response to neuronal activity.90 miR-132 exerts its anti-inflammatory effect by targeting acetylcholinesterase.91 miRNA dysfunction leads to neurodegenerative diseases and acute neurological injury.92 In hippocampal neurons, the downregulation of miRNA-132 results in decreased dendritic arborization, while miRNA-132 overexpression increases the likelihood of synaptic transmission.93 These studies show that miRNA-132 is one of the essential regulators of synaptic plasticity.
Neural differentiation is also triggered by miRNA-9 from embryonic stem cells.94 miRNA-9 has a pro-differentiation role, i.e., miRNA-9 targets forkhead box protein G1 (FoxG1) expression by Cajal–Retzius neuronal differentiation.49 Moreover, miRNA-9 increases the axonal projection and differentiation of spinal motoneurons by targeting forkhead box protein P1 (FoxP1).95 miRNA-9 also restricts the expression of forkhead box protein P2 (FoxP2) and triggers the maturation and migration of cortical neurons.96 miRNA-9/9 in cooperation with miRNA-124 can amplify the crucial role of miRNA-9 in neuronal differentiation by transforming human fibroblast cells into functional neurons.97
An important miRNA in neuronal plasticity is miRNA-134 which is shown to be localized to the synapse in the hippocampal neurons of rats. miRNA-134 is regulated by the protein deacetylase SIRT1. Knockdown of SIRT1 results in elevating miRNA-134 expression as well as decreasing CREB and synaptic plasticity.98
One of the brain miRNAs that play a pro-differentiation role is miRNA-137 which is found in the neural stem cells of embryonic99 and adult mouse brains.100 High miR-137 expressions increase neural differentiation and reduce neural stem cell proliferation. A miRNA that is specifically expressed in mammalian brains is miRNA-219. This miRNA is essential and adequate for increasing the differentiation of oligodendrocytes in the mouse by inhibiting negative regulators of oligodendrocyte differentiation.101
Studies have demonstrated that miRNAs are an important reason for neurodevelopmental, neuropsychiatric, and neurodegenerative diseases.102 Evidence suggests that a change in miRNA-137 expression levels influences functional connectivity and brain activation in individuals with the risk of schizophrenia.103 The pathological importance of miRNA-124 and miRNA-9 and their target genes in schizophrenia disorder deserves further investigation.104 Studies show that the expression of miRNA-132 is considerably reduced in the brains of patients with schizophrenia.105
In the study on the patients with schizophrenia, bipolar disorder, and psychiatrically normal control subjects, compared to the control group in the schizophrenia patient group, all miRNAs reduced expression and exhibited lower expression in the bipolar disorder patient group. These miRNAs include miRNA-425, miRNA-33, miRNA-106b, miRNA-138, miRNA-22, miRNA-151, miRNA-210, miRNA-324-5p, miRNA-338, miRNA-339, and miRNA-15a.106
Profiles of miRNA expression in the brains of old and young mice revealed three miRNAs called miRNA-34, miRNA-124, and let-7, which were changed as a function of age.107 Evidence of increased miR-34a expression in the brain, as well as the blood of older mice, could be a potential marker of brain aging in mice, implying that miRNAs can serve as a suitable biomarker of brain aging in humans.108 Specifically, the study showed that miRNA-124, miRNA-181a, miRNA-9, miRNA-125a, and miRNA-29 changed with aging in the brain.67
Human organs and estrogen synthesis, distribution, and receptors
Estrogen synthesis, in addition to the ovaries, is seen in different parts of the brain, adipose tissue, Vascular endothelium, and muscular cells as well as osteoblasts.109
Which produces estrogen in these tissues depends on the availability of these tissues to C19 steroid precursors.
also, the amount of estrogen metabolism in extra gonadal tissues can be affected by different distributions of enzymes. with age, the secretion and plasma concentration of estrogen decreases significantly.
The presence of large amounts of DHEA and DHEAS in the circulation indicates the presence of a large reservoir of estrogen and testosterone precursors in extra genital tissues (Figure 2).
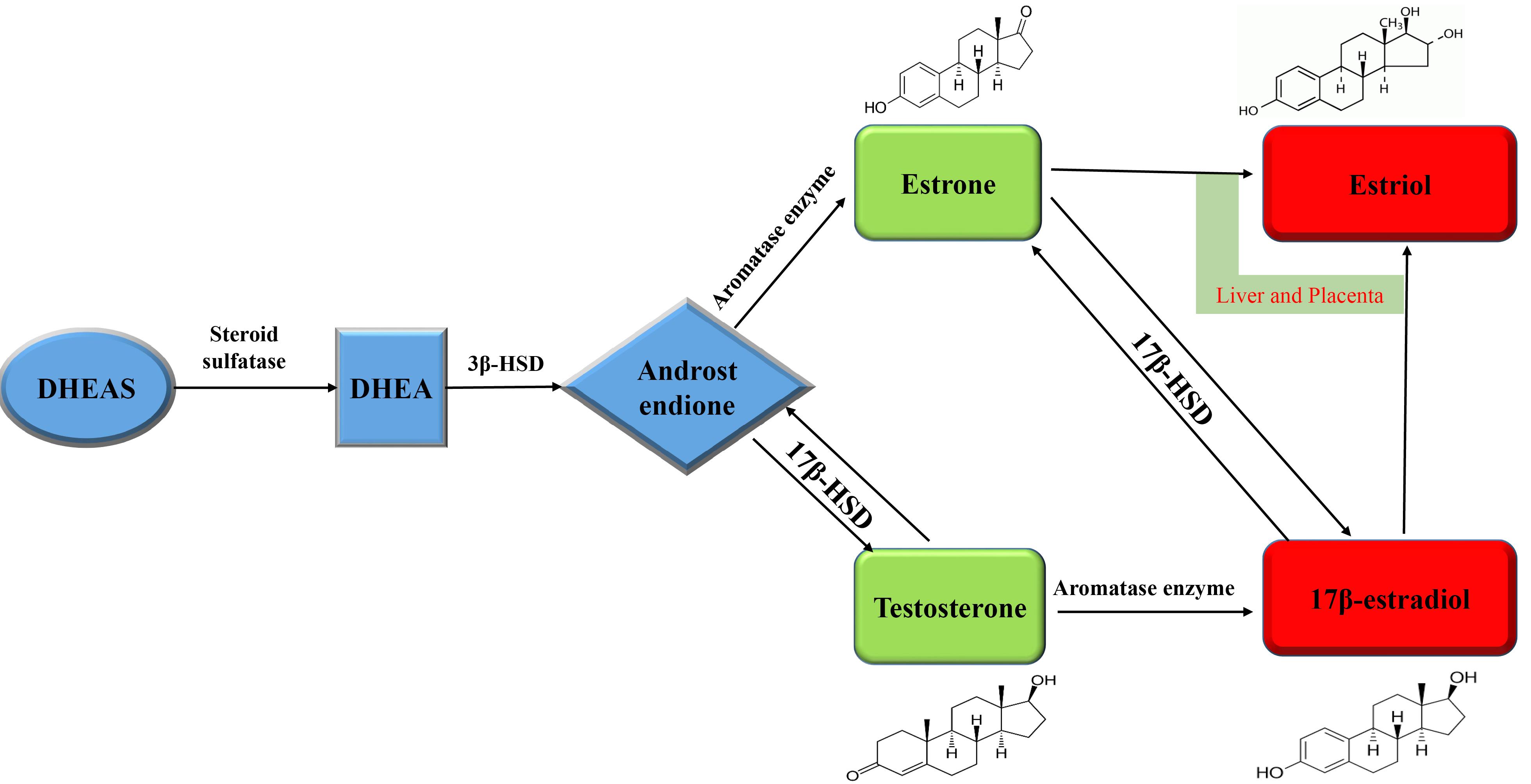
Figure 2.
Schematic of estrogen synthesis. DHEAS – dehydroepiandrosterone sulfate; DHEA – dehydroepiandrosterone; 3β-HSD – 3β-hydroxysteroid dehydrogenase; 17β-HSD – 17β- hydroxysteroid dehydrogenase
.
Schematic of estrogen synthesis. DHEAS – dehydroepiandrosterone sulfate; DHEA – dehydroepiandrosterone; 3β-HSD – 3β-hydroxysteroid dehydrogenase; 17β-HSD – 17β- hydroxysteroid dehydrogenase
Estrogens exert all their effects through their three known receptors. The type of alpha (ERα) before the other receptors was discovered and investigated and with a significant interval after that, the type of beta (ERβ) was discovered with distinct roles.110 The third type of receptor is a G protein-coupled estrogen receptor 1 (GPER1). ERβ at least five and ERα has at least three different isoforms. ERα36 isoform is able to interact with GPER1.
ERβ isoforms due to the inability of transcriptional activity, dimerize with ERα and silencing ERα signaling. Estrogen signaling can be greatly affected by these isoform differences.74,111 The response to different ligands depends on the concentration of the ERα and ERβ and their expression is different in various organs and tissues.112 GPER1 is a plasma membrane receptor that is responsible for estrogen signaling quickly and is expressed independently of other receptors.111 has a high affinity for estrogen, especially 17β-estradiol and activates adenylate cyclase, and finally increasing levels of cAMP.113-115 other steroid hormones have very little tendency to bind to the GPER1.116,117
Comprehensive role of estrogen in health and neuroprotection
Three major endogenous estrogens in females include 17β-estradiol (E2), estrone (E1), and estriol. E2 is the predominant estrogen in humans, the main estrogen in premenopausal women (menopausal transition) that is synthesized from cholesterol in the ovary in response to luteinizing hormone (LH) secretion. In postmenopausal women, E1 is the major estrogen, which is synthesized from adrenal androgens in adipose tissue. Furthermore, E1 and E2 can be synthesized in special tissues, for example, in the breast118 and lung.119 These hormones regulate development, fertility, homeostasis, survival, and proliferation in multiple tissues like the cardiovascular system, brain, skin, breast, bone, intense, lung, and reproductive tract in both men and women.120
Estrogens originate from cholesterol and, in the mitochondria, are converted into 17α-hydroxypregnenolone. In the next step, they are transferred to the smooth endoplasmic reticulum to form dehydroepiandrosterone and, after that, to androgens like testosterone and androstenedione.121 Postmenopausal women seem to be at higher risk for some diseases like heart disease, dementia, and osteoporosis because of a lower level of estrogen.122 Shreds of evidence suggest that all the enzymes required for E2 production are available in the brain and help region-specific estrogen production.123
With the involvement of the aromatase (ARO) enzyme, estrogens are produced from androgens in the CNS of men and women.124 For the final conversion of testosterone into E2 and androstenedione into estrone, a key enzyme is involved which is cytochrome P450. Studies have shown that ARO affects the GnRH/LH surge and produces local estrogen in females; it can also affect neuronal plasticity in the amygdala and hippocampus.125
Estrogen receptors do their function through two main pathways: classical (genomic) or non-classical (non-genomic or rapid signaling). Estrogen in the classical signaling pathway initiates transcription in the target gene by binding to an estrogen receptor (ER), normally ERα or ERβ, which translocates the nucleus and binds to an estrogen response element (ERE). Both ERα and ERβ are extensively expressed normally in all cell types overall the brain, for example, endothelial cells, glia, neurons, and areas that are sensitive to ischemia like the hippocampus and neocortex.126
Moreover, non-classical estrogen signaling is through G-protein coupled estrogen receptor 1 (GPER1), which is located on intracellular and plasma membranes and has been identified to mediate neuroprotective and anti-apoptotic effects in the hippocampal and cortical neurons of mice.127,128
Recent studies have represented that estrogen provides benefits that positively influence acute injury and plasticity as well as regeneration of neurons after ischemia.129 It can improve memory function in animals receiving estrogen supplementation after ischemia.130 Estrogen triggers a cascade of subcellular actions after an ischemic injury, which includes both genomic and non-genomic mechanisms like stabilizing the blood-brain barrier,131 reducing the brain edema,132 vasculature dilating, increasing cerebral blood flow, inflammation suppressing,133 and upregulating cell survival mediators.134 Moreover, estrogen has an antioxidant effect and prevents lipid peroxidation.135
Activating the receptor of N-methyl-D-aspartate may be involved in neuroprotection via estrogen. Estrogen,136 like estrone and estradiol, may be able to improve antioxidant effects in human neuroblastoma cells (SHSY5Y) and neuronal cultures via increasing ATP levels, mitochondrial membrane potential (MMP), and manganese superoxide dismutase (MnSOD) activity,137 which is a very important modulator of the brain metabolic system and may contribute to processes in all the bioenergetic system including metabolism and glucose transport, as well as mitochondrial respiration and ATP production.138 Nowadays, it is recognized that the nuclear transcription of different proteins can be regulated by estrogen which affects the function of mitochondria such as nuclear respiratory factor-1 and peroxisome proliferator-activated receptor-gamma coactivator-1.139 Therefore, this function is crucial for activating genes encoding proteins, that are involved in the mitochondrial electron transport chain complexes and mitochondrial biogenesis.
Menopause is a period of women’s life that influences their physiological and psychological conditions and brings some problems for their quality of life.140 The levels of sex hormone-like estrogen and progesterone decrease in this part of their life. Menopause has some symptoms, one of which is memory problems and cognitive impairment.141
It is recognized that during menopause, the antioxidant enzyme systems (e.g., superoxide dismutase and glutathione peroxidase), and antioxidant vitamins (e.g., vitamin C and vitamin E) are reduced, which is associated with the high level of oxidative stress.142 In the nervous system tissue, estrogen limits cell death by decreasing elevated intracellular Ca2+. This element has an important role in extending ischemic damage induced by ROS. Antioxidant effects of estrogen hinder neurodegeneration, particularly in the hippocampus through different mechanisms,143 This could clarify how estrogen improves cognitive functions.144
The formation of new synaptic connections is another effect of estrogen on the CNS.145 OVX due to estrogen deficiency affects learning and memory.146 Estrogen can increase the amount of brain-derived neurotrophic factor (BDNF) in the CNS, specifically the hippocampus.147 It can be effective in the formation of synapses by altering ERαs expression.148-150 because it has been demonstrated that ERs regulate synaptic protein levels in CA3 and CA1 areas in the hippocampus of humans.151
Estrogen activates some anti-inflammatory factors like GPER1. GPER1 inhibits nuclear factor kappa beta (NF-κB) and its target genes (e.g., TNFα and IL-1β) that lead to neuroprotection in rats with cerebral ischemia.152 In addition, activated GPER1 via estrogen can improve the function of tight junctions, and reduce the permeability of the blood-brain barrier.153 The timing and dose of estrogen therapy are really important for achieving the required neuroprotective effects.154 Neuroinflammation parameters that increase in ovariectomized mice hippocampus, like microglia activation and NF-κB, are also estimated for discovering mood disorders.155
Furthermore, the anti-inflammatory effect of estrogen is essential for CNS development (proliferation or differentiation of neural stem/progenitor cells)156 and important for synaptic plasticity regulation.157
Researchers have also recognized the effects of estrogen on modulating mood in females.158 Mood disorders increase during menopause in women because of estrogen reduction and two predominant symptoms are depression and anxiety.159 The low estrogen levels at the diestrus stage lead to depression-like behaviors in rodents,160 estrogen deficiency can also cause depressive behaviors in ovariectomized rats.161
Estrogen’s multifaceted impact on miRNA regulation and brain function
Even though the number of investigations in miRNAs in animals and humans has increased rapidly in these years, there are still a few studies on miRNA regulation by estrogen. Some steroid hormones, like testosterone, progesterone, and estradiol, regulate miRNA expression.162 As we know, miRNAs have their own promoter elements or are placed on their host genes. It is proven that E2 signaling changes the expression of Dicer, Drosha, and Argonaute and, thus, can regulate the maturation of miRNA.163 Furthermore, sex hormones regulate miRNAs directly via binding to the promoter of miRNA elements or indirectly via binding to nuclear hormone receptors. Consequently, this ligand binding to the nuclear receptor regulates multiple gene transcription and/or recruits coactivators or corepressors.164
There is a different sex-specific model of miRNA expression in the brains of rodents. Blocking the conversion of testosterone into estradiol in men causes this pattern to disappear, showing that miRNAs are regulated by estrogens.165 miRNA-23a expression is also dependent on sex differences and its effect can be related to the sex-specific activation of cell death in a cerebral ischemia model.166 Furthermore, miRNA-101a and miRNA-199a are shown to be the regulators of COX-2 in the developing preoptic area, which is induced by E2 and is required to differentiate the density of the dendritic spine in this region in different sexes.167
Taken together, all these studies demonstrate that tissue-specific miRNAs can be regulated in a differential procedure throughout the development process in the brains of males and females. Consequently, the changes in gene and protein expression can alter the outcomes of some diseases that are associated with sex differences, like stroke. Experimental studies show that some members of the miRNA-200 family such as miRNA200b-3P, miRNA-141-3p, miRNA-429, and miRNA200a-3P have a sex-specific expression pattern in cortex regions of the brain. This study showed that male rats had lower levels of these miRNAs than females in the P0 area, but in the P7 area, females had lower levels of miRNAs than males and adults.168
MiRNAs regulate cell processes that are important for brain sexual differentiation. For instance, the anteroventral periventricular nucleus in the brain has a bigger size in females than in males.169 It means that E2 activates the apoptotic pathways via miRNA regulation.170 Most of the miRNAs are involved in regulating the apoptotic process.171
The gonadal steroid hormone E2 plays an important role in regulating biochemical processing and miRNA expression in some systems, such as models of cancer cells.172 Sufficient evidence has shown that miRNAs respond to estrogen. A study on zebrafish showed that microRNA can be regulated by estrogen.173 which is cell type-specific, e.g. miRNA-196b that targets Hoxb8a directly and is increased with E2 in the skin but is down-regulated in the liver and intestines.174 Moreover, E2 can regulate miRNA expression differently in abnormal and normal tissue; for example, E2 downregulates the expression of miRNA-26 in leiomyoma cells but increases this miRNA in myometrial cells.175
The let-7 miRNA family is up-regulated by estradiol.176 Estrogen reduces proliferation and increases neuronal differentiation in neuroblastoma via let-7.177
Research has shown that E2 has a neuroprotective effect by reducing the time of recovery after the stroke and improving memory and learning in aged humans, rodents, and primates.178
Increasing age causes worldwide changes in the expression of the neuronal gene. Recent investigations have shown that the expression of miRNA changes with age in the aging female brain as a result of decreasing estrogen. Age-related miRNA changes can be responsible for regulating all the changes in gene expression.107
One of the findings from studies has been that E2 can control hippocampal-mediated emotion or stress via differentially regulating miRNA-9 and miRNA-9-3p. They have reported that E2 decreases miRNA-9 expression in the dorsal hippocampus of 3-month-old rats, but increases at the same time miRNA-9-3p expression in animals.179 They have also shown that a subset of miRNAs in the ventral hippocampus like let-7i, miRNA- 495, miRNA-125a, miRNA-9-3p, miRNA-9, miRNA-181a, and miRNA-7a is regulated by E2 in an age-dependent pattern.179
In other studies, 18-month-old female rats are ovariectomized and, then, an acute estradiol treatment is prescribed at 1, 4, 8, or 12 weeks after OVX. Also, the expression of several miRNA processing proteins is measured. Taken together, their results- in the aged hypothalamus- reveal that long ovarian hormone deprivation changes the E2 regulation of mature miRNAs. Analysis of this study suggests that E2 raises the expression of mature miRNA-9, miRNA-7a, let-7i, miRNA-181a, and miRNA-9-3p one week after OVX. However E2 treatment decreases miRNA-495 levels at 12 weeks after OVX. Analyses have demonstrated that six out of seven miRNAs such as let-7i, miRNA-495, miRNA-125a, miRNA-181a, miRNA-9-3p, and miRNA-9 are increased as the result of age alone. Specifically, other results show that expression of let-7i increases at 12 weeks after OVX in vehicle-treated animals, and miRNA-181a, miRNA-9-3p, miRNA-125a, miRNA-495, and miRNA-9 increase in both vehicles- and E2-treated animals at 8 and 12 weeks after OVX. Recent studies have demonstrated that this miRNA plays a substantial role in brain and nervous system development180 and has tumor-suppressor-like properties in glioblastomas.181
Overall insights into estrogen signaling and epigenetic regulation
We have two types of estrogen-dependent signaling. Genomic and non-genomic and the estrogen-ER complex to DNA can be done both directly and indirectly. The direct pathway is the usual signaling path that estrogen-ER complex acts as a transcriptional activator promoting gene expression.182 In the indirect type of signaling path, ligand-activated ERs bind to DNA through protein-protein interactions with other classes of transcription factors.183
Non-genomic effects of E2 usually occur within seconds.184 This fast activity is created by an ‘orphan’ GPR30 or plasma membrane (PM)-associated ER.185 Molecular mechanisms of the non-genomic signaling depend on the variety of cells, numerous, diverse, and activated with various protein-kinase cascades. It is also associated with the type of cellular receptor (ERα, ERβ, and GPER1). ERs can also be activated via the general Phosphorylation state of the receptors such as protein kinase C or protein kinase A, in the absence of 17β-estradiol or another suitable ligand. this way is called the Ligand-independent signaling method. Studies have shown that conjugates with E2 initiate intracellular kinase cascade activities including PI3K/AKT and MAPK/ERK rapidly.186-188
in recent years, another group of important regulators of gene expression has received special attention as epigenetic mechanisms, including the methylation of miRNA, DNA, and histone modifications. Most posttranslational modifications of histone proteins including phosphorylation, deamination, acetylation, ubiquitination, and methylation are dynamic.189,190 Estrogen signaling in addition to gene activation. In addition to coactivators, corepressors are also very effective in suppressing and activating genes involved in the estrogen signaling pathway.74,110 Generally, chromatin modifications are necessary for estrogen-mediated transcriptional gene expression. Epigenetic mechanisms have an important role in the regulation of estrogen signaling and the control of ER gene expression. Similarly, miRNAs such as miR-22, miR-18a, miR-221/222, and miR-206 were also implicated in ER gene expression in normal and tumorous cells.191,192 Estrogen signaling is also able to regulate the expression of miRNAs and certain chromatin-modifying enzymes.174 Finally, miRNAs identified as targeting GPER, and ER are respectively, miR-424 and miR-92.193,194
Studies have shown that exposure to different doses of estrogen causes hypomethylation or hyper-methylation of genes, resulting in suppression or activation of certain genes.195-198 Generally epigenetic regulation and Estrogen signaling are two regulatory mechanisms essential for tissue homeostasis.
Estrogenic regulation of miRNAs for neuroprotection in major brain disorders
Estrogen has neuroprotective effects on various major diseases and conditions, like Alzheimer’s disease (AD),199 Parkinson’s disease,200,201,202 multiple sclerosis (MS),203 spinal cord injury,204 ischemic stroke.205 and retinal degeneration.206 The human lifespan is increasing with each generation due to an increase in age-related disorders. Studies have shown that E2 of less than normal levels is one of the reasons that cause women after menopause to have risks of acquiring mild cognitive impairments, dementia, and AD. 206
Studies have suggested that estrogen has neuroprotective roles against AD-related pathology; these beneficial effects are directly connected to decreased amyloid-β (Aβ) peptides and pathologic aggregates of tau protein,207 which is one of the properties of this hormone. Estrogen therapy decreases Aβ-induced neuronal death via ERα-dependent signaling pathways.208 Researchers have also recognized that estrogen receptors can regulate the expression of miRNAs that are involved in modulating the phosphorylation state of tau. ERα and ERβ have reverse effects on regulating miRNA-218/ protein tyrosine phosphatase α (PTPα) signaling. miRNA-218 targets PTPα and reduces its levels with increasing tau phosphorylation. ERα and ERβ are shown to increase and attenuate miRNA-218 levels in HEK293/tau cells overexpressing ERα or ERβ, respectively.209 miRNA-218 can be expressed in the hippocampus210 and is activated during neuronal differentiation.211
Another miRNA that is involved in AD is miRNA-106a/b which is expressed abundantly in the hippocampus.68 It is recognized that there is an opposite relationship between the miRNA-106a expression and STAT3 in the hippocampus of ovariectomized mice. Moreover, miRNA-106a is able to inhibit directly the expression of STAT3. Hence, estrogen can indirectly upregulate miRNA-106a in mice 12 weeks after OVX. miRNA-106 regulates negatively cholesterol efflux via the target gene ATP-binding cassette TransporterA1(ABCA1) as a therapeutic target for AD212 and may help regulate β-amyloid precursor protein (APP) expression in the brain and differentiate the neurons.213 Increasing APP protein levels cause high levels of Aβ and, consequently, synaptic dysfunction, neurodegeneration, and cognitive impairment.214
MS is one of the CNS inflammatory autoimmune disorders, a classical autoimmune disease that results in injury to oligodendrocytes, loss of myelin, and neurological dysfunction.215 Studies have clarified selective estrogen-receptor modulators increase CNS remyelination independent of estrogen receptors.216 Previous studies have revealed six miRNAs (miR-422a, miR-572, miR-1826, miR-614, miR-648, miR-1826, miR-22, and miR-614) that are implicated in MS.217 Among these miRNAs, miR-22 may inhibit estrogen signaling by targeting the estrogen receptor alpha mRNA. However, the estrogen-MS signaling remains unclear.218
CNS injuries like traumatic brain injury, acute ischemic stroke, and SCI increase the risk of death and disability throughout the world.219 Studies further elucidate that progesterone and estrogen play a key role in neuroprotection after ischemia in ovariectomized female animals.220 Furthermore, in male rats with diabetes, estrogen reduces ischemic stroke infarct size.221 miRNA dysregulation may contribute to the outcome after stroke.222 Estrogen also suppresses NF-κB activation via controlling miRNA-125b and let-7a that regulate expression KappaB-Ras2 levels.223 Recent studies show that inhibiting miRNA181–5p has protective effects on focal ischemia in the female mouse with focal ischemia, which can be via balancing estrogen receptors present on astrocytes.224 Whilst previous studies have identified several miRNAs, many studies still need to explain the role of estrogen in the regulation of miRNAs in ischemic stroke or other diseases in the brain.
Studies show that in general, the expression of both types of estrogen receptors (ERα and ERβ) increases during neural development,225,226 but after birth, the expression of these receptors decreases in the brain and with a wide distribution pattern. It is limited to certain areas of the brain.227,228 With the onset of old age, systemic and specific changes occur in the brain and the flexibility of the brain against side effects is affected, and changes in the level of estrogen receptors are not excluded from this issue, and it has been shown that there is a significant decrease with age. It is seen in the levels of ERα and ERβ in the synapses of CA1 neurons in the hippocampus of female rats.227,229 Therefore, we will lose a level of neuroprotection during this period. Menopause, we see that it seems that this makes the risk due to the lack of this defense barrier double in old women. For example, memory problems and mood changes after menopause in women, as well as the higher prevalence of Alzheimer's disease in women than in men, are related to estrogen deficiency. Due to the inflammatory nature of Alzheimer's disease, it seems that estrogen levels and increasing the duration of estrogen use may affect the process of Alzheimer's disease through neurotrophic, anti-inflammatory effects, increasing cerebral blood flow and reducing the formation of f3-amyloid and increasing the expression of apolipoprotein E gene. Make it more resistant to the disease process.230-232 Estrogen has also been shown to delay or prevent the onset of Parkinson's disease, and several mechanisms have been proposed in animal models for this, including: the positive and protective effect of estrogen on dopamine neurotransmission as well as the reduction of dopamine reuptake in synapses. Reducing the response threshold of L-DOPA and increasing the sensitivity of neurons to dopamine.233,234 Therefore, it seems that by understanding the spatial and temporal expression and signaling of estrogen receptors, we may gain a new perspective on the pathological and physiological aging of the brain, and the gender differences in this process will also be revealed.
Since brain aging and various brain diseases such as Alzheimer’s etc are thought to result from an imbalance between nerve repair and damage, estrogen seems to play a significant role in slowing down these processes and repair, dendritic branching, and survival. have neurons Past studies have stated various mechanisms for exerting these estrogen effects, including: through a direct effect on estrogen receptors located in neurons of different brain regions and effects such as: increasing the concentration, release, reabsorption and inactivation of neurotransmitters such as dopamine, serotonin and norepinephrine in nerve synapses, increasing the number of receptors of these transmitters,220,232,235,236 and also through binding to endothelium receptors and stimulating the release of nitric oxide, which causes vasodilation and increases blood supply to the brain (which is the brain for metabolism It is very dependent on this blood flow) and prevents plaque formation and finally leads to its own protective and neurotrophic effects in the brain.233,237-239
Conclusion
Recent studies show that miRNAs have emerged as novel regulatory mechanisms in pathological and physiological processes by modulating gene expression profiles at the post-transcriptional level, and miRNAs have also been shown to be potential tools for prognosis or diagnosis. and biomarkers are non-invasive and cost-effective and have provided insights into brain function. However, knowledge about the regulatory effects of estrogen on miRNA profiles in brain function is still scarce. Therefore, new therapeutic approaches attempt to identify new miRNAs. In the pathogenesis of diseases, they have been prescribed or manipulated, among which the importance of estrogen in brain function can also be considered in miRNA-based treatments.
Competing Interests
The authors declare no potential competing interest.
Ethical Approval
Not applicable.
Acknowledgements
I would like to thank the members of this research group for their efforts and contributions. This research received no external funding. all authors have agreed on the submission of this article and declare that they have no conflicts of interest also all of them have been involved in writing and editing the article.
References
- Whiteford HA, Degenhardt L, Rehm J, Baxter AJ, Ferrari AJ, Erskine HE. Global burden of disease attributable to mental and substance use disorders: findings from the Global Burden of Disease Study 2010. Lancet 2013; 382(9904):1575-86. doi: 10.1016/s0140-6736(13)61611-6 [Crossref] [ Google Scholar]
- Brown DC, Hayward MD, Montez JK, Hummer RA, Chiu CT, Hidajat MM. The significance of education for mortality compression in the United States. Demography 2012; 49(3):819-40. doi: 10.1007/s13524-012-0104-1 [Crossref] [ Google Scholar]
- Maki PM, Henderson VW. Cognition and the menopause transition. Menopause 2016; 23(7):803-5. doi: 10.1097/gme.0000000000000681 [Crossref] [ Google Scholar]
- Taylor CM, Pritschet L, Yu S, Jacobs EG. Applying a women’s health lens to the study of the aging brain. Front Hum Neurosci 2019; 13:224. doi: 10.3389/fnhum.2019.00224 [Crossref] [ Google Scholar]
- Luine V, Frankfurt M. Interactions between estradiol, BDNF and dendritic spines in promoting memory. Neuroscience 2013; 239:34-45. doi: 10.1016/j.neuroscience.2012.10.019 [Crossref] [ Google Scholar]
- Amar L, Benoit C, Beaumont G, Vacher CM, Crepin D, Taouis M. MicroRNA expression profiling of hypothalamic arcuate and paraventricular nuclei from single rats using Illumina sequencing technology. J Neurosci Methods 2012; 209(1):134-43. doi: 10.1016/j.jneumeth.2012.05.033 [Crossref] [ Google Scholar]
- Rahimian R, Cordeau P Jr, Kriz J. Brain response to injuries: when microglia go sexist. Neuroscience 2019; 405:14-23. doi: 10.1016/j.neuroscience.2018.02.048 [Crossref] [ Google Scholar]
- Kireev RA, Vara E, Viña J, Tresguerres JA. Melatonin and oestrogen treatments were able to improve neuroinflammation and apoptotic processes in dentate gyrus of old ovariectomized female rats. Age (Dordr) 2014; 36(5):9707. doi: 10.1007/s11357-014-9707-3 [Crossref] [ Google Scholar]
- Lu Y, An Y, Lv C, Ma W, Xi Y, Xiao R. Dietary soybean isoflavones in Alzheimer’s disease prevention. Asia Pac J Clin Nutr 2018; 27(5):946-54. doi: 10.6133/apjcn.052018.01 [Crossref] [ Google Scholar]
- Cordeau P Jr, Lalancette-Hébert M, Weng YC, Kriz J. Estrogen receptors alpha mediates postischemic inflammation in chronically estrogen-deprived mice. Neurobiol Aging 2016; 40:50-60. doi: 10.1016/j.neurobiolaging.2016.01.002 [Crossref] [ Google Scholar]
- Liu S, Jiang T, Zhong Y, Yu Y. miR-210 inhibits cell migration and invasion by targeting the brain-derived neurotrophic factor in glioblastoma. J Cell Biochem 2019; 120(7):11375-82. doi: 10.1002/jcb.28414 [Crossref] [ Google Scholar]
- Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 2004; 116(2):281-97. doi: 10.1016/s0092-8674(04)00045-5 [Crossref] [ Google Scholar]
- Catalanotto C, Cogoni C, Zardo G. MicroRNA in control of gene expression: an overview of nuclear functions. Int J Mol Sci 2016; 17(10):1712. doi: 10.3390/ijms17101712 [Crossref] [ Google Scholar]
- Swarbrick S, Wragg N, Ghosh S, Stolzing A. Systematic review of miRNA as biomarkers in Alzheimer’s disease. Mol Neurobiol 2019; 56(9):6156-67. doi: 10.1007/s12035-019-1500-y [Crossref] [ Google Scholar]
- Ambros V. The evolution of our thinking about microRNAs. Nat Med 2008; 14(10):1036-40. doi: 10.1038/nm1008-1036 [Crossref] [ Google Scholar]
- Ruvkun G. The perfect storm of tiny RNAs. Nat Med 2008; 14(10):1041-5. doi: 10.1038/nm1008-1041 [Crossref] [ Google Scholar]
- Baulcombe DC. Short silencing RNA: the dark matter of genetics?. Cold Spring HarbSymp Quant Biol 2006; 71:13-20. doi: 10.1101/sqb.2006.71.052 [Crossref] [ Google Scholar]
- Couzin J. Couzin JGeneticsErasing microRNAs reveals their powerful punch. Science 2007; 316(5824):530. doi: 10.1126/science.316.5824.530 [Crossref] [ Google Scholar]
- Berkhout B, Jeang KT. RISCy business: microRNAs, pathogenesis, and viruses. J Biol Chem 2007; 282(37):26641-5. doi: 10.1074/jbc.R700023200 [Crossref] [ Google Scholar]
- Vasudevan S, Tong Y, Steitz JA. Switching from repression to activation: microRNAs can up-regulate translation. Science 2007; 318(5858):1931-4. doi: 10.1126/science.1149460 [Crossref] [ Google Scholar]
- Saliminejad K, Khorram Khorshid HR, Soleymani Fard S, Ghaffari SH. An overview of microRNAs: Biology, functions, therapeutics, and analysis methods. J Cell Physiol 2019; 234(5):5451-65. doi: 10.1002/jcp.27486 [Crossref] [ Google Scholar]
- Lionetti M, Agnelli L, Mosca L, Fabris S, Andronache A, Todoerti K. Integrative high-resolution microarray analysis of human myeloma cell lines reveals deregulated miRNA expression associated with allelic imbalances and gene expression profiles. Genes Chromosomes Cancer 2009; 48(6):521-31. doi: 10.1002/gcc.20660 [Crossref] [ Google Scholar]
- Esquela-Kerscher A, Slack FJ. Oncomirs - microRNAs with a role in cancer. Nat Rev Cancer 2006; 6(4):259-69. doi: 10.1038/nrc1840 [Crossref] [ Google Scholar]
- Kozomara A, Birgaoanu M, Griffiths-Jones S. miRBase: from microRNA sequences to function. Nucleic Acids Res 2019; 47(D1):D155-62. doi: 10.1093/nar/gky1141 [Crossref] [ Google Scholar]
- Meng Y, Shao C, Wang H, Chen M. Are all the miRBase-registered microRNAs true? A structure- and expression-based re-examination in plants. RNA Biol 2012; 9(3):249-53. doi: 10.4161/rna.19230 [Crossref] [ Google Scholar]
- Brown M, Suryawanshi H, Hafner M, Farazi TA, Tuschl T. Mammalian miRNA curation through next-generation sequencing. Front Genet 2013; 4:145. doi: 10.3389/fgene.2013.00145 [Crossref] [ Google Scholar]
- Griffiths-Jones S. The microRNA registry. Nucleic Acids Res 2004; 32(Database issue):D109-11. doi: 10.1093/nar/gkh023 [Crossref] [ Google Scholar]
- Backes C, Meder B, Hart M, Ludwig N, Leidinger P, Vogel B. Prioritizing and selecting likely novel miRNAs from NGS data. Nucleic Acids Res 2016; 44(6):e53. doi: 10.1093/nar/gkv1335 [Crossref] [ Google Scholar]
- Fromm B, Billipp T, Peck LE, Johansen M, Tarver JE, King BL. A uniform system for the annotation of vertebrate microRNA genes and the evolution of the human microRNAome. Annu Rev Genet 2015; 49:213-42. doi: 10.1146/annurev-genet-120213-092023 [Crossref] [ Google Scholar]
- Alles J, Fehlmann T, Fischer U, Backes C, Galata V, Minet M. An estimate of the total number of true human miRNAs. Nucleic Acids Res 2019; 47(7):3353-64. doi: 10.1093/nar/gkz097 [Crossref] [ Google Scholar]
- Nilsen TW. Mechanisms of microRNA-mediated gene regulation in animal cells. Trends Genet 2007; 23(5):243-9. doi: 10.1016/j.tig.2007.02.011 [Crossref] [ Google Scholar]
- Wu L, Belasco JG. Let me count the ways: mechanisms of gene regulation by miRNAs and siRNAs. Mol Cell 2008; 29(1):1-7. doi: 10.1016/j.molcel.2007.12.010 [Crossref] [ Google Scholar]
- Pillai RS, Bhattacharyya SN, Artus CG, Zoller T, Cougot N, Basyuk E. Inhibition of translational initiation by let-7 microRNA in human cells. Science 2005; 309(5740):1573-6. doi: 10.1126/science.1115079 [Crossref] [ Google Scholar]
- Xie M, Li M, Vilborg A, Lee N, Shu MD, Yartseva V. Mammalian 5’-capped microRNA precursors that generate a single microRNA. Cell 2013; 155(7):1568-80. doi: 10.1016/j.cell.2013.11.027 [Crossref] [ Google Scholar]
- Kim YK, Kim B, Kim VN. Re-evaluation of the roles of DROSHA, export in 5, and DICER in microRNA biogenesis. Proc Natl Acad Sci U S A 2016; 113(13):E1881-9. doi: 10.1073/pnas.1602532113 [Crossref] [ Google Scholar]
- Nguyen TA, Park J, Dang TL, Choi YG, Kim VN. Microprocessor depends on hemin to recognize the apical loop of primary microRNA. Nucleic Acids Res 2018; 46(11):5726-36. doi: 10.1093/nar/gky248 [Crossref] [ Google Scholar]
- Abdelfattah AM, Park C, Choi MY. Update on non-canonical microRNAs. Biomol Concepts 2014; 5(4):275-87. doi: 10.1515/bmc-2014-0012 [Crossref] [ Google Scholar]
- Guenther UP, Weinberg DE, Zubradt MM, Tedeschi FA, Stawicki BN, Zagore LL. The helicase Ded1p controls use of near-cognate translation initiation codons in 5’ UTRs. Nature 2018; 559(7712):130-4. doi: 10.1038/s41586-018-0258-0 [Crossref] [ Google Scholar]
- Ha M, Kim VN. Regulation of microRNA biogenesis. Nat Rev Mol Cell Biol 2014; 15(8):509-24. doi: 10.1038/nrm3838 [Crossref] [ Google Scholar]
- Zealy RW, Wrenn SP, Davila S, Min KW, Yoon JH. microRNA-binding proteins: specificity and function. Wiley Interdiscip Rev RNA 2017; 8(5):e1414. doi: 10.1002/wrna.1414 [Crossref] [ Google Scholar]
- Guo H, Ingolia NT, Weissman JS, Bartel DP. Mammalian microRNAs predominantly act to decrease target mRNA levels. Nature 2010; 466(7308):835-40. doi: 10.1038/nature09267 [Crossref] [ Google Scholar]
- Lang MF, Yang S, Zhao C, Sun G, Murai K, Wu X. Genome-wide profiling identified a set of miRNAs that are differentially expressed in glioblastoma stem cells and normal neural stem cells. PLoS One 2012; 7(4):e36248. doi: 10.1371/journal.pone.0036248 [Crossref] [ Google Scholar]
- Scarisbrick IA. Methods and Materials for Modulating Resistance to Apoptosis Using KLK6 Antisense and MIRNA Molecules. Google Patents; 2019. Available from: https://patents.google.com/patent/US10279037B2/en.
- Hammond SM. An overview of microRNAs. Adv Drug Deliv Rev 2015; 87:3-14. doi: 10.1016/j.addr.2015.05.001 [Crossref] [ Google Scholar]
- Bader AG. miR-34 - a microRNA replacement therapy is headed to the clinic. Front Genet 2012; 3:120. doi: 10.3389/fgene.2012.00120 [Crossref] [ Google Scholar]
- Montgomery RL, Yu G, Latimer PA, Stack C, Robinson K, Dalby CM. MicroRNA mimicry blocks pulmonary fibrosis. EMBO Mol Med 2014; 6(10):1347-56. doi: 10.15252/emmm.201303604 [Crossref] [ Google Scholar]
- Lehrbach NJ, Armisen J, Lightfoot HL, Murfitt KJ, Bugaut A, Balasubramanian S. LIN-28 and the poly(U) polymerase PUP-2 regulate let-7 microRNA processing in Caenorhabditis elegans. Nat Struct Mol Biol 2009; 16(10):1016-20. doi: 10.1038/nsmb.1675 [Crossref] [ Google Scholar]
- Jin XF, Wu N, Wang L, Li J. Circulating microRNAs: a novel class of potential biomarkers for diagnosing and prognosing central nervous system diseases. Cell Mol Neurobiol 2013; 33(5):601-13. doi: 10.1007/s10571-013-9940-9 [Crossref] [ Google Scholar]
- Shibata M, Kurokawa D, Nakao H, Ohmura T, Aizawa S. MicroRNA-9 modulates Cajal-Retzius cell differentiation by suppressing Foxg1 expression in mouse medial pallium. J Neurosci 2008; 28(41):10415-21. doi: 10.1523/jneurosci.3219-08.2008 [Crossref] [ Google Scholar]
- Das E, Bhattacharyya NP. MicroRNA-432 contributes to dopamine cocktail and retinoic acid induced differentiation of human neuroblastoma cells by targeting NESTIN and RCOR1 genes. FEBS Lett 2014; 588(9):1706-14. doi: 10.1016/j.febslet.2014.03.015 [Crossref] [ Google Scholar]
- Sasidharan V, Marepally S, Elliott SA, Baid S, Lakshmanan V, Nayyar N. The miR-124 family of microRNAs is crucial for regeneration of the brain and visual system in the planarian Schmidteamediterranea. Development 2017; 144(18):3211-23. doi: 10.1242/dev.144758 [Crossref] [ Google Scholar]
- Gu X, Xu X, Jia C, Wang J, Zhang J, Gao Q. Molecular mechanisms involved in the regulation of neurodevelopment by miR-124. Mol Neurobiol 2023; 60(7):3569-83. doi: 10.1007/s12035-023-03271-5 [Crossref] [ Google Scholar]
- You Q, Gong Q, Han YQ, Pi R, Du YJ, Dong SZ. Role of miR-124 in the regulation of retinoic acid-induced Neuro-2a cell differentiation. Neural Regen Res 2020; 15(6):1133-9. doi: 10.4103/1673-5374.270417 [Crossref] [ Google Scholar]
- Barzegar Behrooz A, Latifi-Navid H, Nezhadi A, Świat M, Los M, Jamalpoor Z. Molecular mechanisms of microRNAs in glioblastoma pathogenesis. BiochimBiophys Acta Mol Cell Res 2023; 1870(6):119482. doi: 10.1016/j.bbamcr.2023.119482 [Crossref] [ Google Scholar]
- Lanza M, Cuzzocrea S, Oddo S, Esposito E, Casili G. The role of miR-128 in neurodegenerative diseases. Int J Mol Sci 2023; 24(7):6024. doi: 10.3390/ijms24076024 [Crossref] [ Google Scholar]
- Lippi G, Steinert JR, Marczylo EL, D’Oro S, Fiore R, Forsythe ID. Targeting of the Arpc3 actin nucleation factor by miR-29a/b regulates dendritic spine morphology. J Cell Biol 2011; 194(6):889-904. doi: 10.1083/jcb.201103006 [Crossref] [ Google Scholar]
- Improta-Caria AC, Nonaka CK, Cavalcante BR, De Sousa RAL, Aras Júnior R, de Freitas Souza BS. Modulation of microRNAs as a potential molecular mechanism involved in the beneficial actions of physical exercise in Alzheimer disease. Int J Mol Sci 2020; 21(14):4977. doi: 10.3390/ijms21144977 [Crossref] [ Google Scholar]
- Brain O, Owens BM, Pichulik T, Allan P, Khatamzas E, Leslie A. The intracellular sensor NOD2 induces microRNA-29 expression in human dendritic cells to limit IL-23 release. Immunity 2013; 39(3):521-36. doi: 10.1016/j.immuni.2013.08.035 [Crossref] [ Google Scholar]
- Subramanian M, Mills WT 4th, Paranjpe MD, Onuchukwu US, Inamdar M, Maytin AR. Growth-suppressor microRNAs mediate synaptic overgrowth and behavioral deficits in fragile X mental retardation protein deficiency. iScience 2024; 27(1):108676. doi: 10.1016/j.isci.2023.108676 [Crossref] [ Google Scholar]
- Hansen KL. MiR-132 as a Dynamic Regulator of Neuronal Structure and Cognitive Capacity [dissertation]. The Ohio State University; 2015.
- Meng Q, Ye C, Lu Y. miR-181c regulates ischemia/reperfusion injury-induced neuronal cell death by regulating c-Fos signaling. Pharmazie 2020; 75(2):90-3. doi: 10.1691/ph.2020.9856 [Crossref] [ Google Scholar]
- Inouye MO, Colameo D, Ammann I, Winterer J, Schratt G. miR-329- and miR-495-mediated Prr7 down-regulation is required for homeostatic synaptic depression in rat hippocampal neurons. Life Sci Alliance 2022; 5(12):e202201520. doi: 10.26508/lsa.202201520 [Crossref] [ Google Scholar]
- Balan S, Toyoshima M, Yoshikawa T. Contribution of induced pluripotent stem cell technologies to the understanding of cellular phenotypes in schizophrenia. Neurobiol Dis 2019; 131:104162. doi: 10.1016/j.nbd.2018.04.021 [Crossref] [ Google Scholar]
- Hu YB, Zhang YF, Wang H, Ren RJ, Cui HL, Huang WY. miR-425 deficiency promotes necroptosis and dopaminergic neurodegeneration in Parkinson’s disease. Cell Death Dis 2019; 10(8):589. doi: 10.1038/s41419-019-1809-5 [Crossref] [ Google Scholar]
- Shi L, Cheng Z, Zhang J, Li R, Zhao P, Fu Z. hsa-mir-181a and hsa-mir-181b function as tumor suppressors in human glioma cells. Brain Res 2008; 1236:185-93. doi: 10.1016/j.brainres.2008.07.085 [Crossref] [ Google Scholar]
- Xu W, Zhao M, Lin Z, Liu H, Ma H, Hong Q. Increased expression of plasma hsa-miR-181a in male patients with heroin addiction use disorder. J Clin Lab Anal 2020; 34(11):e23486. doi: 10.1002/jcla.23486 [Crossref] [ Google Scholar]
- Somel M, Guo S, Fu N, Yan Z, Hu HY, Xu Y. MicroRNA, mRNA, and protein expression link development and aging in human and macaque brain. Genome Res 2010; 20(9):1207-18. doi: 10.1101/gr.106849.110 [Crossref] [ Google Scholar]
- Camkurt MA, Karababa İF, Erdal ME, Kandemir SB, Fries GR, Bayazıt H. MicroRNA dysregulation in manic and euthymic patients with bipolar disorder. J Affect Disord 2020; 261:84-90. doi: 10.1016/j.jad.2019.09.060 [Crossref] [ Google Scholar]
- Wang H, Liu J, Zong Y, Xu Y, Deng W, Zhu H. miR-106b aberrantly expressed in a double transgenic mouse model for Alzheimer’s disease targets TGF-β type II receptor. Brain Res 2010; 1357:166-74. doi: 10.1016/j.brainres.2010.08.023 [Crossref] [ Google Scholar]
- Lou YL, Guo F, Liu F, Gao FL, Zhang PQ, Niu X. miR-210 activates notch signaling pathway in angiogenesis induced by cerebral ischemia. Mol Cell Biochem 2012; 370(1-2):45-51. doi: 10.1007/s11010-012-1396-6 [Crossref] [ Google Scholar]
- Zaccagnini G, Greco S, Voellenkle C, Gaetano C, Martelli F. miR-210 hypoxamiR in angiogenesis and diabetes. Antioxid Redox Signal 2022; 36(10-12):685-706. doi: 10.1089/ars.2021.0200 [Crossref] [ Google Scholar]
- Xu HS, Zong HL, Shang M, Ming X, Zhao JP, Ma C. MiR-324-5p inhibits proliferation of glioma by target regulation of GLI1. Eur Rev Med Pharmacol Sci 2014; 18(6):828-32. [ Google Scholar]
- Daswani RR. Mir-138 Controls Hippocampal Interneuron Function and Short-Term Memory [dissertation]. ETH Zurich; 2021. 10.3929/ethz-b-000505199.
- Heldring N, Pike A, Andersson S, Matthews J, Cheng G, Hartman J. Estrogen receptors: how do they signal and what are their targets. Physiol Rev 2007; 87(3):905-31. doi: 10.1152/physrev.00026.2006 [Crossref] [ Google Scholar]
- Zhang Y, Wei G, Di Z, Zhao Q. miR-339-5p inhibits alcohol-induced brain inflammation through regulating NF-κB pathway. BiochemBiophys Res Commun 2014; 452(3):450-6. doi: 10.1016/j.bbrc.2014.08.092 [Crossref] [ Google Scholar]
- Amar L, Benoit C, Beaumont G, Vacher CM, Crepin D, Taouis M. MicroRNA expression profiling of hypothalamic arcuate and paraventricular nuclei from single rats using Illumina sequencing technology. J Neurosci Methods 2012; 209(1):134-43. doi: 10.1016/j.jneumeth.2012.05.033 [Crossref] [ Google Scholar]
- Delaloy C, Liu L, Lee JA, Su H, Shen F, Yang GY. MicroRNA-9 coordinates proliferation and migration of human embryonic stem cell-derived neural progenitors. Cell Stem Cell 2010; 6(4):323-35. doi: 10.1016/j.stem.2010.02.015 [Crossref] [ Google Scholar]
- Schratt GM, Tuebing F, Nigh EA, Kane CG, Sabatini ME, Kiebler M. A brain-specific microRNA regulates dendritic spine development. Nature 2006; 439(7074):283-9. doi: 10.1038/nature04367 [Crossref] [ Google Scholar]
- Jovičić A, Roshan R, Moisoi N, Pradervand S, Moser R, Pillai B. Comprehensive expression analyses of neural cell-type-specific miRNAs identify new determinants of the specification and maintenance of neuronal phenotypes. J Neurosci 2013; 33(12):5127-37. doi: 10.1523/jneurosci.0600-12.2013 [Crossref] [ Google Scholar]
- He M, Liu Y, Wang X, Zhang Michael Q, Hannon GJ, Huang ZJ. Cell-Type-Based Analysis of MicroRNA Profiles in the Mouse Brain. Neuron 2012; 73(1):35-48. doi: 10.1016/j.neuron.2011.11.010 [Crossref] [ Google Scholar]
- Kye MJ, Liu T, Levy SF, Xu NL, Groves BB, Bonneau R. Somatodendritic microRNAs identified by laser capture and multiplex RT-PCR. RNA 2007; 13(8):1224-34. doi: 10.1261/rna.480407 [Crossref] [ Google Scholar]
- Yu JY, Chung KH, Deo M, Thompson RC, Turner DL. MicroRNA miR-124 regulates neurite outgrowth during neuronal differentiation. Exp Cell Res 2008; 314(14):2618-33. doi: 10.1016/j.yexcr.2008.06.002 [Crossref] [ Google Scholar]
- Samaraweera L, Grandinetti KB, Huang R, Spengler BA, Ross RA. MicroRNAs define distinct human neuroblastoma cell phenotypes and regulate their differentiation and tumorigenicity. BMC Cancer 2014; 14:309. doi: 10.1186/1471-2407-14-309 [Crossref] [ Google Scholar]
- Meng Y, Shang F, Zhu Y. miR-124 participates in the proliferation and differentiation of brain glioma stem cells through regulating Nogo/NgR expression. Exp Ther Med 2019; 18(4):2783-8. doi: 10.3892/etm.2019.7914 [Crossref] [ Google Scholar]
- Franke K, Otto W, Johannes S, Baumgart J, Nitsch R, Schumacher S. miR-124-regulated RhoG reduces neuronal process complexity via ELMO/Dock180/Rac1 and Cdc42 signalling. EMBO J 2012; 31(13):2908-21. doi: 10.1038/emboj.2012.130 [Crossref] [ Google Scholar]
- Sanuki R, Onishi A, Koike C, Muramatsu R, Watanabe S, Muranishi Y. miR-124a is required for hippocampal axogenesis and retinal cone survival through Lhx2 suppression. Nat Neurosci 2011; 14(9):1125-34. doi: 10.1038/nn.2897 [Crossref] [ Google Scholar]
- Gao C, Shen J, Meng ZX, He XF. Sevoflurane inhibits glioma cells proliferation and metastasis through miRNA-124-3p/ROCK1 axis. Pathol Oncol Res 2020; 26(2):947-54. doi: 10.1007/s12253-019-00597-1 [Crossref] [ Google Scholar]
- Rajasethupathy P, Fiumara F, Sheridan R, Betel D, Puthanveettil SV, Russo JJ. Characterization of small RNAs in Aplysia reveals a role for miR-124 in constraining synaptic plasticity through CREB. Neuron 2009; 63(6):803-17. doi: 10.1016/j.neuron.2009.05.029 [Crossref] [ Google Scholar]
- Edbauer D, Neilson JR, Foster KA, Wang CF, Seeburg DP, Batterton MN. Regulation of synaptic structure and function by FMRP-associated microRNAs miR-125b and miR-132. Neuron 2010; 65(3):373-84. doi: 10.1016/j.neuron.2010.01.005 [Crossref] [ Google Scholar]
- Wayman GA, Davare M, Ando H, Fortin D, Varlamova O, Cheng HY. An activity-regulated microRNA controls dendritic plasticity by down-regulating p250GAP. Proc Natl Acad Sci U S A 2008; 105(26):9093-8. doi: 10.1073/pnas.0803072105 [Crossref] [ Google Scholar]
- Taouis M. MicroRNAs in the hypothalamus. Best Pract Res Clin Endocrinol Metab 2016; 30(5):641-51. doi: 10.1016/j.beem.2016.11.006 [Crossref] [ Google Scholar]
- Saugstad JA. MicroRNAs as effectors of brain function with roles in ischemia and injury, neuroprotection, and neurodegeneration. J Cereb Blood Flow Metab 2010; 30(9):1564-76. doi: 10.1038/jcbfm.2010.101 [Crossref] [ Google Scholar]
- Lambert TJ, Storm DR, Sullivan JM. MicroRNA132 modulates short-term synaptic plasticity but not basal release probability in hippocampal neurons. PLoS One 2010; 5(12):e15182. doi: 10.1371/journal.pone.0015182 [Crossref] [ Google Scholar]
- Saunders LR, Sharma AD, Tawney J, Nakagawa M, Okita K, Yamanaka S. miRNAs regulate SIRT1 expression during mouse embryonic stem cell differentiation and in adult mouse tissues. Aging (Albany NY) 2010; 2(7):415-31. doi: 10.18632/aging.100176 [Crossref] [ Google Scholar]
- Otaegi G, Pollock A, Hong J, Sun T. MicroRNA miR-9 modifies motor neuron columns by a tuning regulation of FoxP1 levels in developing spinal cords. J Neurosci 2011; 31(3):809-18. doi: 10.1523/jneurosci.4330-10.2011 [Crossref] [ Google Scholar]
- Clovis YM, Enard W, Marinaro F, Huttner WB, De Pietri Tonelli D. Convergent repression of Foxp2 3’UTR by miR-9 and miR-132 in embryonic mouse neocortex: implications for radial migration of neurons. Development 2012; 139(18):3332-42. doi: 10.1242/dev.078063 [Crossref] [ Google Scholar]
- Yoo AS, Sun AX, Li L, Shcheglovitov A, Portmann T, Li Y. MicroRNA-mediated conversion of human fibroblasts to neurons. Nature 2011; 476(7359):228-31. doi: 10.1038/nature10323 [Crossref] [ Google Scholar]
- Gao J, Wang WY, Mao YW, Gräff J, Guan JS, Pan L. A novel pathway regulates memory and plasticity via SIRT1 and miR-134. Nature 2010; 466(7310):1105-9. doi: 10.1038/nature09271 [Crossref] [ Google Scholar]
- Kallert SM, Darbre S, Bonilla WV, Kreutzfeldt M, Page N, Müller P. Replicating viral vector platform exploits alarmin signals for potent CD8 + T cell-mediated tumour immunotherapy. Nat Commun 2017; 8:15327. doi: 10.1038/ncomms15327 [Crossref] [ Google Scholar]
- Silber J, Lim DA, Petritsch C, Persson AI, Maunakea AK, Yu M. miR-124 and miR-137 inhibit proliferation of glioblastoma multiforme cells and induce differentiation of brain tumor stem cells. BMC Med 2008; 6:14. doi: 10.1186/1741-7015-6-14 [Crossref] [ Google Scholar]
- Zhao X, He X, Han X, Yu Y, Ye F, Chen Y. MicroRNA-mediated control of oligodendrocyte differentiation. Neuron 2010; 65(5):612-26. doi: 10.1016/j.neuron.2010.02.018 [Crossref] [ Google Scholar]
- Olde Loohuis NF, Kos A, Martens GJ, Van Bokhoven H, Nadif Kasri N, Aschrafi A. MicroRNA networks direct neuronal development and plasticity. Cell Mol Life Sci 2012; 69(1):89-102. doi: 10.1007/s00018-011-0788-1 [Crossref] [ Google Scholar]
- Whalley HC, Papmeyer M, Romaniuk L, Sprooten E, Johnstone EC, Hall J. Impact of a microRNA MIR137 susceptibility variant on brain function in people at high genetic risk of schizophrenia or bipolar disorder. Neuropsychopharmacology 2012; 37(12):2720-9. doi: 10.1038/npp.2012.137 [Crossref] [ Google Scholar]
- Beveridge NJ, Gardiner E, Carroll AP, Tooney PA, Cairns MJ. Schizophrenia is associated with an increase in cortical microRNA biogenesis. Mol Psychiatry 2010; 15(12):1176-89. doi: 10.1038/mp.2009.84 [Crossref] [ Google Scholar]
- Miller BH, Zeier Z, Xi L, Lanz TA, Deng S, Strathmann J. MicroRNA-132 dysregulation in schizophrenia has implications for both neurodevelopment and adult brain function. Proc Natl Acad Sci U S A 2012; 109(8):3125-30. doi: 10.1073/pnas.1113793109 [Crossref] [ Google Scholar]
- Moreau MP, Bruse SE, David-Rus R, Buyske S, Brzustowicz LM. Altered microRNA expression profiles in postmortem brain samples from individuals with schizophrenia and bipolar disorder. Biol Psychiatry 2011; 69(2):188-93. doi: 10.1016/j.biopsych.2010.09.039 [Crossref] [ Google Scholar]
- Inukai S, de Lencastre A, Turner M, Slack F. Novel microRNAs differentially expressed during aging in the mouse brain. PLoS One 2012; 7(7):e40028. doi: 10.1371/journal.pone.0040028 [Crossref] [ Google Scholar]
- Li X, Khanna A, Li N, Wang E. Circulatory miR34a as an RNAbased, noninvasive biomarker for brain aging. Aging (Albany NY) 2011; 3(10):985-1002. doi: 10.18632/aging.100371 [Crossref] [ Google Scholar]
- Simpson ER. Sources of estrogen and their importance. J Steroid Biochem Mol Biol 2003; 86(3-5):225-30. doi: 10.1016/s0960-0760(03)00360-1 [Crossref] [ Google Scholar]
- Nilsson S, Mäkelä S, Treuter E, Tujague M, Thomsen J, Andersson G. Mechanisms of estrogen action. Physiol Rev 2001; 81(4):1535-65. doi: 10.1152/physrev.2001.81.4.1535 [Crossref] [ Google Scholar]
- Xiao X, Wu ZC, Chou KC. A multi-label classifier for predicting the subcellular localization of gram-negative bacterial proteins with both single and multiple sites. PLoS One 2011; 6(6):e20592. doi: 10.1371/journal.pone.0020592 [Crossref] [ Google Scholar]
- Nilsson S, Gustafsson JÅ. Estrogen receptors: therapies targeted to receptor subtypes. Clin PharmacolTher 2011; 89(1):44-55. doi: 10.1038/clpt.2010.226 [Crossref] [ Google Scholar]
- Belcher SM, Le HH, Spurling L, Wong JK. Rapid estrogenic regulation of extracellular signal- regulated kinase 1/2 signaling in cerebellar granule cells involves a G protein- and protein kinase A-dependent mechanism and intracellular activation of protein phosphatase 2A. Endocrinology 2005; 146(12):5397-406. doi: 10.1210/en.2005-0564 [Crossref] [ Google Scholar]
- Filardo E, Quinn J, Pang Y, Graeber C, Shaw S, Dong J. Activation of the novel estrogen receptor G protein-coupled receptor 30 (GPR30) at the plasma membrane. Endocrinology 2007; 148(7):3236-45. doi: 10.1210/en.2006-1605 [Crossref] [ Google Scholar]
- Funakoshi T, Yanai A, Shinoda K, Kawano MM, Mizukami Y. G protein-coupled receptor 30 is an estrogen receptor in the plasma membrane. BiochemBiophys Res Commun 2006; 346(3):904-10. doi: 10.1016/j.bbrc.2006.05.191 [Crossref] [ Google Scholar]
- Filardo EJ, Thomas P. Minireview: G protein-coupled estrogen receptor-1, GPER-1: its mechanism of action and role in female reproductive cancer, renal and vascular physiology. Endocrinology 2012; 153(7):2953-62. doi: 10.1210/en.2012-1061 [Crossref] [ Google Scholar]
- Sandén C, Broselid S, Cornmark L, Andersson K, Daszkiewicz-Nilsson J, Mårtensson UE. G protein-coupled estrogen receptor 1/G protein-coupled receptor 30 localizes in the plasma membrane and traffics intracellularly on cytokeratin intermediate filaments. Mol Pharmacol 2011; 79(3):400-10. doi: 10.1124/mol.110.069500 [Crossref] [ Google Scholar]
- Bondesson M, Hao R, Lin CY, Williams C, Gustafsson JÅ. Estrogen receptor signaling during vertebrate development. BiochimBiophys Acta 2015; 1849(2):142-51. doi: 10.1016/j.bbagrm.2014.06.005 [Crossref] [ Google Scholar]
- Heilbroner SP, Xanthopoulos EP, Buono D, Huang Y, Carrier D, Shah A. Impact of estrogen monotherapy on survival in women with stage III-IV non-small cell lung cancer. Lung Cancer 2019; 129:8-15. doi: 10.1016/j.lungcan.2018.12.021 [Crossref] [ Google Scholar]
- Viña J, Sastre J, Pallardó FV, Gambini J, Borrás C. Modulation of longevity-associated genes by estrogens or phytoestrogens. Biol Chem 2008; 389(3):273-7. doi: 10.1515/bc.2008.027 [Crossref] [ Google Scholar]
- BA G, Melmed S, Polonsky KS, Larsen PR, Kronenberg HM (eds), Williams Textbook of Endocrinology, 13th edn. Elsevier 2016. Reproduced with permission of Elsevier. Male Sexual Dysfunction: A Clinical Guide, 2017, 30.
- Dalal PK, Agarwal M. Postmenopausal syndrome. Indian J Psychiatry 2015; 57(Suppl 2):S222-32. doi: 10.4103/0019-5545.161483 [Crossref] [ Google Scholar]
- Rossetti MF, Cambiasso MJ, Holschbach MA, Cabrera R. Oestrogens and progestagens: synthesis and action in the brain. J Neuroendocrinol 2016;28(7). 10.1111/jne.12402.
- Li J, Gibbs RB. Detection of estradiol in rat brain tissues: contribution of local versus systemic production. Psychoneuroendocrinology 2019; 102:84-94. doi: 10.1016/j.psyneuen.2018.11.037 [Crossref] [ Google Scholar]
- Bender RA, Zhou L, Vierk R, Brandt N, Keller A, Gee CE. Sex-dependent regulation of aromatase-mediated synaptic plasticity in the basolateral amygdala. J Neurosci 2017; 37(6):1532-45. doi: 10.1523/jneurosci.1532-16.2016 [Crossref] [ Google Scholar]
- Kritzer MF. Regional, laminar, and cellular distribution of immunoreactivity for ER alpha and ER beta in the cerebral cortex of hormonally intact, adult male and female rats. Cereb Cortex 2002; 12(2):116-28. doi: 10.1093/cercor/12.2.116 [Crossref] [ Google Scholar]
- Kajta M, Litwa E, Rzemieniec J, Wnuk A, Lason W, Zelek-Molik A. Isomer-nonspecific action of dichlorodiphenyltrichloroethane on aryl hydrocarbon receptor and G-protein-coupled receptor 30 intracellular signaling in apoptotic neuronal cells. Mol Cell Endocrinol 2014; 392(1-2):90-105. doi: 10.1016/j.mce.2014.05.008 [Crossref] [ Google Scholar]
- Wnuk A, Kajta M. Steroid and xenobiotic receptor signalling in apoptosis and autophagy of the nervous system. Int J Mol Sci 2017; 18(11):2394. doi: 10.3390/ijms18112394 [Crossref] [ Google Scholar]
- Gibbs RB. Estrogen therapy and cognition: a review of the cholinergic hypothesis. Endocr Rev 2010; 31(2):224-53. doi: 10.1210/er.2009-0036 [Crossref] [ Google Scholar]
- Gulinello M, Lebesgue D, Jover-Mengual T, Zukin RS, Etgen AM. Acute and chronic estradiol treatments reduce memory deficits induced by transient global ischemia in female rats. HormBehav 2006; 49(2):246-60. doi: 10.1016/j.yhbeh.2005.07.010 [Crossref] [ Google Scholar]
- Walf AA, Frye CA. A review and update of mechanisms of estrogen in the hippocampus and amygdala for anxiety and depression behavior. Neuropsychopharmacology 2006; 31(6):1097-111. doi: 10.1038/sj.npp.1301067 [Crossref] [ Google Scholar]
- Yuen N, Anderson SE, Glaser N, Tancredi DJ, O’Donnell ME. Cerebral blood flow and cerebral edema in rats with diabetic ketoacidosis. Diabetes 2008; 57(10):2588-94. doi: 10.2337/db07-1410 [Crossref] [ Google Scholar]
- Liu R, Wen Y, Perez E, Wang X, Day AL, Simpkins JW. 17beta-estradiol attenuates blood-brain barrier disruption induced by cerebral ischemia-reperfusion injury in female rats. Brain Res 2005; 1060(1-2):55-61. doi: 10.1016/j.brainres.2005.08.048 [Crossref] [ Google Scholar]
- Fujita K, Kato T, Shibayama K, Imada H, Yamauchi M, Yoshimoto N. Protective effect against 17beta-estradiol on neuronal apoptosis in hippocampus tissue following transient ischemia/recirculation in mongolian gerbils via down-regulation of tissue transglutaminase activity. Neurochem Res 2006; 31(8):1059-68. doi: 10.1007/s11064-006-9114-y [Crossref] [ Google Scholar]
- Azcoitia I, Barreto GE, Garcia-Segura LM. Molecular mechanisms and cellular events involved in the neuroprotective actions of estradiol Analysis of sex differences. Front Neuroendocrinol 2019; 55:100787. doi: 10.1016/j.yfrne.2019.100787 [Crossref] [ Google Scholar]
- Connell BJ, Crosby KM, Richard MJ, Mayne MB, Saleh TM. Estrogen-mediated neuroprotection in the cortex may require NMDA receptor activation. Neuroscience 2007; 146(1):160-9. doi: 10.1016/j.neuroscience.2007.01.014 [Crossref] [ Google Scholar]
- Grimm A, Schmitt K, Lang UE, Mensah-Nyagan AG, Eckert A. Improvement of neuronal bioenergetics by neurosteroids: implications for age-related neurodegenerative disorders. BiochimBiophys Acta 2014; 1842(12 Pt A):2427-38. doi: 10.1016/j.bbadis.2014.09.013 [Crossref] [ Google Scholar]
- Ngo ST, Steyn FJ, McCombe PA. Gender differences in autoimmune disease. Front Neuroendocrinol 2014; 35(3):347-69. doi: 10.1016/j.yfrne.2014.04.004 [Crossref] [ Google Scholar]
- Klinge CM. Estrogens regulate life and death in mitochondria. J BioenergBiomembr 2017; 49(4):307-24. doi: 10.1007/s10863-017-9704-1 [Crossref] [ Google Scholar]
- Ansar S, Alhefdhi T, Aleem AM. Status of trace elements and antioxidants in premenopausal and postmenopausal phase of life: a comparative study. Int J Clin Exp Med 2015; 8(10):19486-90. [ Google Scholar]
- Pompili A, Arnone B, D’Amico M, Federico P, Gasbarri A. Evidence of estrogen modulation on memory processes for emotional content in healthy young women. Psychoneuroendocrinology 2016; 65:94-101. doi: 10.1016/j.psyneuen.2015.12.013 [Crossref] [ Google Scholar]
- Grimm A, Mensah-Nyagan AG, Eckert A. Alzheimer, mitochondria and gender. NeurosciBiobehav Rev 2016; 67:89-101. doi: 10.1016/j.neubiorev.2016.04.012 [Crossref] [ Google Scholar]
- Sribnick EA, Del Re AM, Ray SK, Woodward JJ, Banik NL. Estrogen attenuates glutamate-induced cell death by inhibiting Ca2 + influx through L-type voltage-gated Ca2 + channels. Brain Res 2009; 1276:159-70. doi: 10.1016/j.brainres.2009.04.022 [Crossref] [ Google Scholar]
- Wang F, Song YF, Yin J, Liu ZH, Mo XD, Wang DG. Spatial memory impairment is associated with hippocampal insulin signals in ovariectomized rats. PLoS One 2014; 9(8):e104450. doi: 10.1371/journal.pone.0104450 [Crossref] [ Google Scholar]
- Sárvári M, Kalló I, Hrabovszky E, Solymosi N, Rodolosse A, Liposits Z. Long-term estrogen receptor beta agonist treatment modifies the hippocampal transcriptome in middle-aged ovariectomized rats. Front Cell Neurosci 2016; 10:149. doi: 10.3389/fncel.2016.00149 [Crossref] [ Google Scholar]
- Engler-Chiurazzi EB, Singh M, Simpkins JW. From the 90’s to now: a brief historical perspective on more than two decades of estrogen neuroprotection. Brain Res 2016; 1633:96-100. doi: 10.1016/j.brainres.2015.12.044 [Crossref] [ Google Scholar]
- Ortiz-Pérez A, Espinosa-Raya J, Picazo O. An enriched environment and 17-beta estradiol produce similar pro-cognitive effects on ovariectomized rats. Cogn Process 2016; 17(1):15-25. doi: 10.1007/s10339-015-0746-1 [Crossref] [ Google Scholar]
- Baumgartner NE, Daniel JM. Estrogen receptor: a critical role in successful female cognitive aging. Climacteric 2021; 24(4):333-9. doi: 10.1080/13697137.2021.1875426 [Crossref] [ Google Scholar]
- Pfaff D, Waters E, Khan Q, Zhang X, Numan M. Minireview: estrogen receptor-initiated mechanisms causal to mammalian reproductive behaviors. Endocrinology 2011; 152(4):1209-17. doi: 10.1210/en.2010-1007 [Crossref] [ Google Scholar]
- Foster TC. Role of estrogen receptor alpha and beta expression and signaling on cognitive function during aging. Hippocampus 2012; 22(4):656-69. doi: 10.1002/hipo.20935 [Crossref] [ Google Scholar]
- Seeley WW, Menon V, Schatzberg AF, Keller J, Glover GH, Kenna H. Dissociable intrinsic connectivity networks for salience processing and executive control. J Neurosci 2007; 27(9):2349-56. doi: 10.1523/jneurosci.5587-06.2007 [Crossref] [ Google Scholar]
- Zhao G, Su Z, Song D, Mao Y, Mao X. The long noncoding RNA MALAT1 regulates the lipopolysaccharide-induced inflammatory response through its interaction with NF-κB. FEBS Lett 2016; 590(17):2884-95. doi: 10.1002/1873-3468.12315 [Crossref] [ Google Scholar]
- Lu D, Qu Y, Shi F, Feng D, Tao K, Gao G. Activation of G protein-coupled estrogen receptor 1 (GPER-1) ameliorates blood-brain barrier permeability after global cerebral ischemia in ovariectomized rats. BiochemBiophys Res Commun 2016; 477(2):209-14. doi: 10.1016/j.bbrc.2016.06.044 [Crossref] [ Google Scholar]
- Manwani B, Liu F, Scranton V, Hammond MD, Sansing LH, McCullough LD. Differential effects of aging and sex on stroke induced inflammation across the lifespan. Exp Neurol 2013; 249:120-31. doi: 10.1016/j.expneurol.2013.08.011 [Crossref] [ Google Scholar]
- Liu T, Ma Y, Zhang R, Zhong H, Wang L, Zhao J. Resveratrol ameliorates estrogen deficiency-induced depression- and anxiety-like behaviors and hippocampal inflammation in mice. Psychopharmacology (Berl) 2019; 236(4):1385-99. doi: 10.1007/s00213-018-5148-5 [Crossref] [ Google Scholar]
- Varshney M, Nalvarte I. Genes, gender, environment, and novel functions of estrogen receptor beta in the susceptibility to neurodevelopmental disorders. Brain Sci 2017; 7(3):24. doi: 10.3390/brainsci7030024 [Crossref] [ Google Scholar]
- Zárate S, Stevnsner T, Gredilla R. Role of estrogen and other sex hormones in brain aging Neuroprotection and DNA Repair. Front Aging Neurosci 2017; 9:430. doi: 10.3389/fnagi.2017.00430 [Crossref] [ Google Scholar]
- Borrow AP, Cameron NM. Estrogenic mediation of serotonergic and neurotrophic systems: implications for female mood disorders. Prog Neuropsychopharmacol Biol Psychiatry 2014; 54:13-25. doi: 10.1016/j.pnpbp.2014.05.009 [Crossref] [ Google Scholar]
- Mulhall S, Andel R, Anstey KJ. Variation in symptoms of depression and anxiety in midlife women by menopausal status. Maturitas 2018; 108:7-12. doi: 10.1016/j.maturitas.2017.11.005 [Crossref] [ Google Scholar]
- Carrier J, Lord C. Could networking and sharing (open) data in an international collaborative effort unravel the mechanisms of sleep disturbances in middle-aged women?. Menopause 2015; 22(7):691-2. doi: 10.1097/gme.0000000000000496 [Crossref] [ Google Scholar]
- Li Y, Li SQ, Gao YM, Li J, Zhang B. Crucial role of Notch signaling in osteogenic differentiation of periodontal ligament stem cells in osteoporotic rats. Cell Biol Int 2014; 38(6):729-36. doi: 10.1002/cbin.10257 [Crossref] [ Google Scholar]
- Klinge CM. miRNAs and estrogen action. Trends Endocrinol Metab 2012; 23(5):223-33. doi: 10.1016/j.tem.2012.03.002 [Crossref] [ Google Scholar]
- Siomi H, Siomi MC. Posttranscriptional regulation of microRNA biogenesis in animals. Mol Cell 2010; 38(3):323-32. doi: 10.1016/j.molcel.2010.03.013 [Crossref] [ Google Scholar]
- Sharma S, Eghbali M. Influence of sex differences on microRNA gene regulation in disease. Biol Sex Differ 2014; 5(1):3. doi: 10.1186/2042-6410-5-3 [Crossref] [ Google Scholar]
- Morgan CP, Bale TL. Early prenatal stress epigenetically programs dysmasculinization in second-generation offspring via the paternal lineage. J Neurosci 2011; 31(33):11748-55. doi: 10.1523/jneurosci.1887-11.2011 [Crossref] [ Google Scholar]
- Siegel C, Li J, Liu F, Benashski SE, McCullough LD. miR-23a regulation of X-linked inhibitor of apoptosis (XIAP) contributes to sex differences in the response to cerebral ischemia. Proc Natl Acad Sci U S A 2011; 108(28):11662-7. doi: 10.1073/pnas.1102635108 [Crossref] [ Google Scholar]
- Chakrabarty A, Tranguch S, Daikoku T, Jensen K, Furneaux H, Dey SK. MicroRNA regulation of cyclooxygenase-2 during embryo implantation. Proc Natl Acad Sci U S A 2007; 104(38):15144-9. doi: 10.1073/pnas.0705917104 [Crossref] [ Google Scholar]
- Murphy SJ, Lusardi TA, Phillips JI, Saugstad JA. Sex differences in microRNA expression during development in rat cortex. Neurochem Int 2014; 77:24-32. doi: 10.1016/j.neuint.2014.06.007 [Crossref] [ Google Scholar]
- Turano A, Osborne BF, Schwarz JM. Sexual differentiation and sex differences in neural development. Curr Top BehavNeurosci 2019; 43:69-110. doi: 10.1007/7854_2018_56 [Crossref] [ Google Scholar]
- Waters EM, Simerly RB. Estrogen induces caspase-dependent cell death during hypothalamic development. J Neurosci 2009; 29(31):9714-8. doi: 10.1523/jneurosci.0135-09.2009 [Crossref] [ Google Scholar]
- Jovanovic M, Hengartner MO. miRNAs and apoptosis: RNAs to die for. Oncogene 2006; 25(46):6176-87. doi: 10.1038/sj.onc.1209912 [Crossref] [ Google Scholar]
- Paris O, Ferraro L, Grober OM, Ravo M, De Filippo MR, Giurato G. Direct regulation of microRNA biogenesis and expression by estrogen receptor beta in hormone-responsive breast cancer. Oncogene 2012; 31(38):4196-206. doi: 10.1038/onc.2011.583 [Crossref] [ Google Scholar]
- Nothnick WB, Healy C. Estrogen induces distinct patterns of microRNA expression within the mouse uterus. Reprod Sci 2010; 17(11):987-94. doi: 10.1177/1933719110377472 [Crossref] [ Google Scholar]
- Cohen A, Shmoish M, Levi L, Cheruti U, Levavi-Sivan B, Lubzens E. Alterations in micro-ribonucleic acid expression profiles reveal a novel pathway for estrogen regulation. Endocrinology 2008; 149(4):1687-96. doi: 10.1210/en.2007-0969 [Crossref] [ Google Scholar]
- Pan Q, Luo X, Chegini N. Differential expression of microRNAs in myometrium and leiomyomas and regulation by ovarian steroids. J Cell Mol Med 2008; 12(1):227-40. doi: 10.1111/j.1582-4934.2007.00207.x [Crossref] [ Google Scholar]
- Bhat-Nakshatri P, Wang G, Collins NR, Thomson MJ, Geistlinger TR, Carroll JS. Estradiol-regulated microRNAs control estradiol response in breast cancer cells. Nucleic Acids Res 2009; 37(14):4850-61. doi: 10.1093/nar/gkp500 [Crossref] [ Google Scholar]
- Perri P, Ponzoni M, Corrias MV, Ceccherini I, Candiani S, Bachetti T. A focus on regulatory networks linking microRNAs, transcription factors and target genes in neuroblastoma. Cancers (Basel) 2021; 13(21):5528. doi: 10.3390/cancers13215528 [Crossref] [ Google Scholar]
- Liu F, Day M, Muñiz LC, Bitran D, Arias R, Revilla-Sanchez R. Activation of estrogen receptor-beta regulates hippocampal synaptic plasticity and improves memory. Nat Neurosci 2008; 11(3):334-43. doi: 10.1038/nn2057 [Crossref] [ Google Scholar]
- Rao YS, Mott NN, Wang Y, Chung WC, Pak TR. MicroRNAs in the aging female brain: a putative mechanism for age-specific estrogen effects. Endocrinology 2013; 154(8):2795-806. doi: 10.1210/en.2013-1230 [Crossref] [ Google Scholar]
- Chen H, Shalom-Feuerstein R, Riley J, Zhang SD, Tucci P, Agostini M. miR-7 and miR-214 are specifically expressed during neuroblastoma differentiation, cortical development and embryonic stem cells differentiation, and control neurite outgrowth in vitro. BiochemBiophys Res Commun 2010; 394(4):921-7. doi: 10.1016/j.bbrc.2010.03.076 [Crossref] [ Google Scholar]
- Kefas B, Godlewski J, Comeau L, Li Y, Abounader R, Hawkinson M. microRNA-7 inhibits the epidermal growth factor receptor and the Akt pathway and is down-regulated in glioblastoma. Cancer Res 2008; 68(10):3566-72. doi: 10.1158/0008-5472.can-07-6639 [Crossref] [ Google Scholar]
- Lodish H, Berk A, Kaiser CA, Kaiser C, Krieger M, Scott MP, et al. Molecular Cell Biology. Macmillan; 2008.
- Björnström L, Sjöberg M. Mechanisms of estrogen receptor signaling: convergence of genomic and nongenomic actions on target genes. Mol Endocrinol 2005; 19(4):833-42. doi: 10.1210/me.2004-0486 [Crossref] [ Google Scholar]
- Chambliss KL, Shaul PW. Estrogen modulation of endothelial nitric oxide synthase. Endocr Rev 2002; 23(5):665-86. doi: 10.1210/er.2001-0045 [Crossref] [ Google Scholar]
- Watson CS, Alyea RA, Jeng YJ, Kochukov MY. Nongenomic actions of low concentration estrogens and xenoestrogens on multiple tissues. Mol Cell Endocrinol 2007; 274(1-2):1-7. doi: 10.1016/j.mce.2007.05.011 [Crossref] [ Google Scholar]
- Razandi M, Pedram A, Levin ER. Plasma membrane estrogen receptors signal to antiapoptosis in breast cancer. Mol Endocrinol 2000; 14(9):1434-47. doi: 10.1210/mend.14.9.0526 [Crossref] [ Google Scholar]
- Jaubert AM, Mehebik-Mojaat N, Lacasa D, Sabourault D, Giudicelli Y, Ribière C. Nongenomic estrogen effects on nitric oxide synthase activity in rat adipocytes. Endocrinology 2007; 148(5):2444-52. doi: 10.1210/en.2006-1329 [Crossref] [ Google Scholar]
- Wang C, Prossnitz ER, Roy SK. G protein-coupled receptor 30 expression is required for estrogen stimulation of primordial follicle formation in the hamster ovary. Endocrinology 2008; 149(9):4452-61. doi: 10.1210/en.2008-0441 [Crossref] [ Google Scholar]
- Gibney ER, Nolan CM. Epigenetics and gene expression. Heredity (Edinb) 2010; 105(1):4-13. doi: 10.1038/hdy.2010.54 [Crossref] [ Google Scholar]
- Portela A, Esteller M. Epigenetic modifications and human disease. Nat Biotechnol 2010; 28(10):1057-68. doi: 10.1038/nbt.1685 [Crossref] [ Google Scholar]
- Adams BD, Furneaux H, White BA. The micro-ribonucleic acid (miRNA) miR-206 targets the human estrogen receptor-alpha (ERalpha) and represses ERalpha messenger RNA and protein expression in breast cancer cell lines. Mol Endocrinol 2007; 21(5):1132-47. doi: 10.1210/me.2007-0022 [Crossref] [ Google Scholar]
- Tang L, Pu Y, Wong DK, Liu T, Tang H, Xiang T. The hepatitis B virus-associated estrogen receptor alpha (ERalpha) was regulated by microRNA-130a in HepG2215 human hepatocellular carcinoma cells. Acta BiochimBiophys Sin (Shanghai) 2011; 43(8):640-6. doi: 10.1093/abbs/gmr051 [Crossref] [ Google Scholar]
- Qian P, Zuo Z, Wu Z, Meng X, Li G, Wu Z. Pivotal role of reduced let-7g expression in breast cancer invasion and metastasis. Cancer Res 2011; 71(20):6463-74. doi: 10.1158/0008-5472.can-11-1322 [Crossref] [ Google Scholar]
- Kim K, Madak-Erdogan Z, Ventrella R, Katzenellenbogen BS. A microRNA196a2* and TP63 circuit regulated by estrogen receptor-α and ERK2 that controls breast cancer proliferation and invasiveness properties. Horm Cancer 2013; 4(2):78-91. doi: 10.1007/s12672-012-0129-3 [Crossref] [ Google Scholar]
- Chao HH, Zhang XF, Chen B, Pan B, Zhang LJ, Li L. Bisphenol A exposure modifies methylation of imprinted genes in mouse oocytes via the estrogen receptor signaling pathway. Histochem Cell Biol 2012; 137(2):249-59. doi: 10.1007/s00418-011-0894-z [Crossref] [ Google Scholar]
- Marques M, Laflamme L, Gaudreau L. Estrogen receptor α can selectively repress dioxin receptor-mediated gene expression by targeting DNA methylation. Nucleic Acids Res 2013; 41(17):8094-106. doi: 10.1093/nar/gkt595 [Crossref] [ Google Scholar]
- Cheng AS, Culhane AC, Chan MW, Venkataramu CR, Ehrich M, Nasir A. Epithelial progeny of estrogen-exposed breast progenitor cells display a cancer-like methylome. Cancer Res 2008; 68(6):1786-96. doi: 10.1158/0008-5472.can-07-5547 [Crossref] [ Google Scholar]
- Ho SM, Tang WY, Belmonte de Frausto J, Prins GS. Developmental exposure to estradiol and bisphenol A increases susceptibility to prostate carcinogenesis and epigenetically regulates phosphodiesterase type 4 variant 4. Cancer Res 2006; 66(11):5624-32. doi: 10.1158/0008-5472.can-06-0516 [Crossref] [ Google Scholar]
- Engler-Chiurazzi EB, Brown CM, Povroznik JM, Simpkins JW. Estrogens as neuroprotectants: estrogenic actions in the context of cognitive aging and brain injury. Prog Neurobiol 2017; 157:188-211. doi: 10.1016/j.pneurobio.2015.12.008 [Crossref] [ Google Scholar]
- Calderón J, Navarro ME, Jimenez-Capdeville ME, Santos-Diaz MA, Golden A, Rodriguez-Leyva I. Exposure to arsenic and lead and neuropsychological development in Mexican children. Environ Res 2001; 85(2):69-76. doi: 10.1006/enrs.2000.4106 [Crossref] [ Google Scholar]
- Rodriguez-Perez AI, Borrajo A, Valenzuela R, Lanciego JL, Labandeira-Garcia JL. Critical period for dopaminergic neuroprotection by hormonal replacement in menopausal rats. Neurobiol Aging 2015; 36(2):1194-208. doi: 10.1016/j.neurobiolaging.2014.10.028 [Crossref] [ Google Scholar]
- Litim N, Morissette M, Di Paolo T. Neuroactive gonadal drugs for neuroprotection in male and female models of Parkinson’s disease. NeurosciBiobehav Rev 2016; 67:79-88. doi: 10.1016/j.neubiorev.2015.09.024 [Crossref] [ Google Scholar]
- Atkinson KC, Lee JB, Hasselmann JPC, Kim SH, Drew A, Soto J. Diffusion tensor imaging identifies aspects of therapeutic estrogen receptor β ligand-induced remyelination in a mouse model of multiple sclerosis. Neurobiol Dis 2019; 130:104501. doi: 10.1016/j.nbd.2019.104501 [Crossref] [ Google Scholar]
- Samantaray S, Das A, Matzelle DC, Yu SP, Wei L, Varma A. Administration of low dose estrogen attenuates persistent inflammation, promotes angiogenesis, and improves locomotor function following chronic spinal cord injury in rats. J Neurochem 2016; 137(4):604-17. doi: 10.1111/jnc.13610 [Crossref] [ Google Scholar]
- Jover-Mengual T, Castelló-Ruiz M, Burguete MC, Jorques M, López-Morales MA, Aliena-Valero A. Molecular mechanisms mediating the neuroprotective role of the selective estrogen receptor modulator, bazedoxifene, in acute ischemic stroke: a comparative study with 17β-estradiol. J Steroid Biochem Mol Biol 2017; 171:296-304. doi: 10.1016/j.jsbmb.2017.05.001 [Crossref] [ Google Scholar]
- Wei Q, Liang X, Peng Y, Yu D, Zhang R, Jin H. 17β-estradiol ameliorates oxidative stress and blue light-emitting diode-induced retinal degeneration by decreasing apoptosis and enhancing autophagy. Drug Des DevelTher 2018; 12:2715-30. doi: 10.2147/dddt.s176349 [Crossref] [ Google Scholar]
- Toro CA, Zhang L, Cao J, Cai D. Sex differences in Alzheimer’s disease: understanding the molecular impact. Brain Res 2019; 1719:194-207. doi: 10.1016/j.brainres.2019.05.031 [Crossref] [ Google Scholar]
- Kim H, Bang OY, Jung MW, Ha SD, Hong HS, Huh K. Neuroprotective effects of estrogen against beta-amyloid toxicity are mediated by estrogen receptors in cultured neuronal cells. Neurosci Lett 2001; 302(1):58-62. doi: 10.1016/s0304-3940(01)01659-7 [Crossref] [ Google Scholar]
- Xiong YS, Liu FF, Liu D, Huang HZ, Wei N, Tan L. Opposite effects of two estrogen receptors on tau phosphorylation through disparate effects on the miR-218/PTPA pathway. Aging Cell 2015; 14(5):867-77. doi: 10.1111/acel.12366 [Crossref] [ Google Scholar]
- Bak M, Silahtaroglu A, Møller M, Christensen M, Rath MF, Skryabin B. MicroRNA expression in the adult mouse central nervous system. RNA 2008; 14(3):432-44. doi: 10.1261/rna.783108 [Crossref] [ Google Scholar]
- Sempere LF, Freemantle S, Pitha-Rowe I, Moss E, Dmitrovsky E, Ambros V. Expression profiling of mammalian microRNAs uncovers a subset of brain-expressed microRNAs with possible roles in murine and human neuronal differentiation. Genome Biol 2004; 5(3):R13. doi: 10.1186/gb-2004-5-3-r13 [Crossref] [ Google Scholar]
- Kim J, Yoon H, Ramírez CM, Lee SM, Hoe HS, Fernández-Hernando C. MiR-106b impairs cholesterol efflux and increases Aβ levels by repressing ABCA1 expression. Exp Neurol 2012; 235(2):476-83. doi: 10.1016/j.expneurol.2011.11.010 [Crossref] [ Google Scholar]
- Patel N, Hoang D, Miller N, Ansaloni S, Huang Q, Rogers JT. MicroRNAs can regulate human APP levels. Mol Neurodegener 2008; 3:10. doi: 10.1186/1750-1326-3-10 [Crossref] [ Google Scholar]
- Bettens K, Brouwers N, Van Miegroet H, Gil A, Engelborghs S, De Deyn PP. Follow-up study of susceptibility loci for Alzheimer’s disease and onset age identified by genome-wide association. J Alzheimers Dis 2010; 19(4):1169-75. doi: 10.3233/jad-2010-1310 [Crossref] [ Google Scholar]
- Crawford AH, Tripathi RB, Foerster S, McKenzie I, Kougioumtzidou E, Grist M. Pre-existing mature oligodendrocytes do not contribute to remyelination following toxin-induced spinal cord demyelination. Am J Pathol 2016; 186(3):511-6. doi: 10.1016/j.ajpath.2015.11.005 [Crossref] [ Google Scholar]
- Rankin KA, Mei F, Kim K, Shen YA, Mayoral SR, Desponts C. Selective estrogen receptor modulators enhance CNS remyelination independent of estrogen receptors. J Neurosci 2019; 39(12):2184-94. doi: 10.1523/jneurosci.1530-18.2019 [Crossref] [ Google Scholar]
- Siegel SR, Mackenzie J, Chaplin G, Jablonski NG, Griffiths L. Circulating microRNAs involved in multiple sclerosis. Mol Biol Rep 2012; 39(5):6219-25. doi: 10.1007/s11033-011-1441-7 [Crossref] [ Google Scholar]
- Pandey DP, Picard D. miR-22 inhibits estrogen signaling by directly targeting the estrogen receptor alpha mRNA. Mol Cell Biol 2009; 29(13):3783-90. doi: 10.1128/mcb.01875-08 [Crossref] [ Google Scholar]
- Herman P, Stein A, Gibbs K, Korsunsky I, Gregersen P, Bloom O. Persons with chronic spinal cord injury have decreased natural killer cell and increased toll-like receptor/inflammatory gene expression. J Neurotrauma 2018; 35(15):1819-29. doi: 10.1089/neu.2017.5519 [Crossref] [ Google Scholar]
- Suzuki S, Gerhold LM, Böttner M, Rau SW, Dela Cruz C, Yang E. Estradiol enhances neurogenesis following ischemic stroke through estrogen receptors alpha and beta. J Comp Neurol 2007; 500(6):1064-75. doi: 10.1002/cne.21240 [Crossref] [ Google Scholar]
- Santizo RA, Xu HL, Ye S, Baughman VL, Pelligrino DA. Loss of benefit from estrogen replacement therapy in diabetic ovariectomized female rats subjected to transient forebrain ischemia. Brain Res 2002; 956(1):86-95. doi: 10.1016/s0006-8993(02)03484-4 [Crossref] [ Google Scholar]
- Dharap A, Bowen K, Place R, Li LC, Vemuganti R. Transient focal ischemia induces extensive temporal changes in rat cerebral microRNAome. J Cereb Blood Flow Metab 2009; 29(4):675-87. doi: 10.1038/jcbfm.2008.157 [Crossref] [ Google Scholar]
- Murphy AJ, Guyre PM, Pioli PA. Estradiol suppresses NF-kappa B activation through coordinated regulation of let-7a and miR-125b in primary human macrophages. J Immunol 2010; 184(9):5029-37. doi: 10.4049/jimmunol.0903463 [Crossref] [ Google Scholar]
- Stary CM, Xu L, Li L, Sun X, Ouyang YB, Xiong X. Inhibition of miR-181a protects female mice from transient focal cerebral ischemia by targeting astrocyte estrogen receptor-α. Mol Cell Neurosci 2017; 82:118-25. doi: 10.1016/j.mcn.2017.05.004 [Crossref] [ Google Scholar]
- Fan X, Xu H, Warner M, Gustafsson JÅ. ERbeta in CNS: new roles in development and function. Prog Brain Res 2010; 181:233-50. doi: 10.1016/s0079-6123(08)81013-8 [Crossref] [ Google Scholar]
- Varshney MK, Inzunza J, Lupu D, Ganapathy V, Antonson P, Rüegg J. Role of estrogen receptor beta in neural differentiation of mouse embryonic stem cells. Proc Natl Acad Sci U S A 2017; 114(48):E10428-e37. doi: 10.1073/pnas.1714094114 [Crossref] [ Google Scholar]
- Mehra RD, Sharma K, Nyakas C, Vij U. Estrogen receptor alpha and beta immunoreactive neurons in normal adult and aged female rat hippocampus: a qualitative and quantitative study. Brain Res 2005; 1056(1):22-35. doi: 10.1016/j.brainres.2005.06.073 [Crossref] [ Google Scholar]
- Kanaya M, Higo S, Ozawa H. Neurochemical characterization of neurons expressing estrogen receptor β in the hypothalamic nuclei of rats using in situ hybridization and immunofluorescence. Int J Mol Sci 2019; 21(1):115. doi: 10.3390/ijms21010115 [Crossref] [ Google Scholar]
- Waters EM, Yildirim M, Janssen WG, Lou WY, McEwen BS, Morrison JH. Estrogen and aging affect the synaptic distribution of estrogen receptor β-immunoreactivity in the CA1 region of female rat hippocampus. Brain Res 2011; 1379:86-97. doi: 10.1016/j.brainres.2010.09.069 [Crossref] [ Google Scholar]
- Uddin MS, Rahman MM, Jakaria M, Rahman MS, Hossain MS, Islam A. Estrogen signaling in Alzheimer’s disease: molecular insights and therapeutic targets for Alzheimer’s dementia. Mol Neurobiol 2020; 57(6):2654-70. doi: 10.1007/s12035-020-01911-8 [Crossref] [ Google Scholar]
- Wang JM, Irwin RW, Brinton RD. Activation of estrogen receptor alpha increases and estrogen receptor beta decreases apolipoprotein E expression in hippocampus in vitro and in vivo. Proc Natl Acad Sci U S A 2006; 103(45):16983-8. doi: 10.1073/pnas.0608128103 [Crossref] [ Google Scholar]
- Russell JK, Jones CK, Newhouse PA. The role of estrogen in brain and cognitive aging. Neurotherapeutics 2019; 16(3):649-65. doi: 10.1007/s13311-019-00766-9 [Crossref] [ Google Scholar]
- Jarras H, Bourque M, Poirier AA, Morissette M, Coulombe K, Di Paolo T. Neuroprotection and immunomodulation of progesterone in the gut of a mouse model of Parkinson’s disease. J Neuroendocrinol 2020; 32(1):e12782. doi: 10.1111/jne.12782 [Crossref] [ Google Scholar]
- Shams WM, Sanio C, Quinlan MG, Brake WG. 17β-estradiol infusions into the dorsal striatum rapidly increase dorsal striatal dopamine release in vivo. Neuroscience 2016; 330:162-70. doi: 10.1016/j.neuroscience.2016.05.049 [Crossref] [ Google Scholar]
- Galea LAM, Frick KM, Hampson E, Sohrabji F, Choleris E. Why estrogens matter for behavior and brain health. NeurosciBiobehav Rev 2017; 76(Pt B):363-79. doi: 10.1016/j.neubiorev.2016.03.024 [Crossref] [ Google Scholar]
- Céspedes Rubio ÁE, Pérez-Alvarez MJ, Lapuente Chala C, Wandosell F. Sex steroid hormones as neuroprotective elements in ischemia models. J Endocrinol 2018; 237(2):R65-81. doi: 10.1530/joe-18-0129 [Crossref] [ Google Scholar]
- Varshney MK, Yu NY, Katayama S, Li X, Liu T, Wu WF. Motor function deficits in the estrogen receptor beta knockout mouse: role on excitatory neurotransmission and myelination in the motor cortex. Neuroendocrinology 2021; 111(1-2):27-44. doi: 10.1159/000506162 [Crossref] [ Google Scholar]
- Villa A, Vegeto E, Poletti A, Maggi A. Estrogens, neuroinflammation, and neurodegeneration. Endocr Rev 2016; 37(4):372-402. doi: 10.1210/er.2016-1007 [Crossref] [ Google Scholar]
- Meyer MR, Barton M. Estrogens and coronary artery disease: new clinical perspectives. Adv Pharmacol 2016; 77:307-60. doi: 10.1016/bs.apha.2016.05.003 [Crossref] [ Google Scholar]